Local temperature near native vascular plants in the Argentine Islands–Kyiv Peninsula region, Antarctic Peninsula: annual variability and approximation using standard meteorological measurements


Опубликована Янв. 1, 2023
Последнее обновление статьи Июль 25, 2023
Abstract
We describe the main features of LT variability that influence native vascular plants in the Antarctic and examine the relationship between the temperature regime at the micro-level and meteorological conditions at the macro-level. We used a period of over a year, during which 37 specialized mini-loggers recorded LT near vascular plants in the Argentine Islands–Kyiv Peninsula region of the Antarctic Peninsula. Rather than measuring standard air or soil temperature, these loggers detect the temperature near the ground, in the microhabitats that harbour vascular plants. On a daily scale, LT correlates with standard (2-m) air temperature, with the values higher at rock slopes than at rock terraces and ledges. A moderate correlation was found with wind and radiation parameters. Seasonality accounted for 75–93% of total LT variability, with better results on open rock terraces compared to protected areas and clefts. LT day-to-day variability during the cold season is mostly responsible for differences in R2 of the annual cycle. We estimated daily mean LT using regression dependencies from 2-m air temperature and wind speed measured at a nearby meteorological station. R2 for these statistical models varies from 0.46 to 0.68. However, they underestimate the observed LT. LT measured on rock slopes showed better modelling results with air temperature, whereas wind speed was a better predictor on rock ledges. This study contributes to our understanding of the micro-scale temperature regime that influences native vascular plants and provides a method for its rough approximation using standard meteorological parameters.
Ключевые слова
Mini-logger, vegetation, Microclimate, microhabitat, Colobanthus quitensis, Deschampsia antarctica
Introduction
Regardless of the distance from inhabited regions, environmental changes in Antarctic terrestrial ecosystems can be good indicators for tracking anthropogenic impacts from global to local scales. This is already clearly evident in the regional climate of the maritime Antarctic (Turner et al. 2005; Turner et al. 2013). Local terrestrial biota, which exist in extremely harsh natural conditions (Hogg et al. 2006; Convey 2012; Cannone et al. 2021), are also sensitive to global climate change (Walther et al. 2002; Aerts et al. 2006; Convey & Smith 2006), even under strong microclimatological influences (Miotke 1988). The necessity for monitoring climate change impacts on vulnerable Antarctic terrestrial ecosystems requires the identification of suitable meteorological parameters for analyses and estimations.
Observations of climate variability in the Antarctic are in most cases carried out at a macro-level, using measurements of air temperature and other meteorological parameters at standard heights above the ground. These measurements are conducted at weather stations that were established to be representative of the state of the climate over a large region (WMO 2010). However, standard meteorological observations often fail to adequately describe the microclimatological conditions in which terrestrial flora, fauna and microbiota are found.
Understanding the relationship between meteorological parameters at micro- and macro-levels is crucial for studying the response to climate change of terrestrial ecosystems and their components in the region. (Here, we define the micro-scale as that of several metres, corresponding to relief microforms like clefts and slopes where terrestrial organisms exist, particularly native vascular plants. Meso- and macro-scales are scales corresponding to standard observations at meteorological stations and represent the weather conditions over the islands where our study took place.) Strong responses to microclimatological conditions have already been shown for vascular plants (Gerighausen et al. 2003; Parnikoza et al. 2009). Numerous factors often cause unpredictable changes in meteorological conditions at the micro-scale (Miotke 1988). As a result, microclimatological conditions within an area as small as a few square metres can be so different that plants growing in one spot in it cannot grow in another (Graae et al. 2012; Convey et al. 2018).
OTCs, which simulate warming in manipulation experiments, are popular equipment for studying microclimatology in the Antarctic. Experimental studies have shown that OTCs had a greater warming effect with distance from the sea, although this effect disappeared when there was strong wind and varied with land-cover type and sunshine duration (Bartak et al. 2019). The temperature regime is also important (Oliphant et al. 2015). Warming in OTCs significantly improves plant development in polar regions by making more liquid water available and increasing photosynthesis (Gjessing & Øvstedal 1989; Bokhorst et al. 2012; Saez et al. 2018; Bartak et al. 2019). Although OTCs are excellent tools for microclimatological studies, some valuable information may be lost during OTC manipulations, hindering our understanding of micro- to macro-scale responses in nature.
Overall, elevation significantly affects meteorological conditions at the micro-level, influencing plant development (Cannone et al. 2016). Permafrost, which occurs in the Argentine Islands at depths of 20–40 cm (Abakumov & Parnikoza 2015; Parnikoza et al. 2016), is another important factor at the micro-scale. The areas with favourable conditions for plant growth are above the permafrost, and individual plants may appear on relief microforms like cracks, elevations and so on (Parnikoza, Berezkina et al. 2018). Favourable weather conditions for plant development are complicated by animal activity (Parnikoza et al. 2015; Cannone et al. 2016), human activity (Tin et al. 2008; Convey 2011) and even the possible impact of different plant species on each other (Molina-Montenegro et al. 2013; Casanova-Katny et al. 2014; Molina-Montenegro et al. 2014).
Meteorological parameters at the micro-scale are affected by seasonal variability and reflect changes in macro-scale conditions (Walton 1982; Guglielmin et al. 2012; Convey et al. 2018). The main drivers of the seasonal thermal regime changes at a micro-scale are the general air temperature variability, solar radiation impacts and snow cover thickness. At a micro-scale, snow cover determines the variation of soil and ground temperature as well as the intensity of cooling during the cold season (Davey et al. 1992).
This present study is based on data from an extensive study area covering a number of islands in the maritime Antarctic. We used measurements of LT on the surface (substrate) in native vascular plants microhabitats in the Argentine Islands–Kyiv Peninsula region, on the Graham Coast of the Antarctic Peninsula (Parnikoza, Miryuta et al. 2018; Savenets et al. 2020). We aimed to detect the most typical temperature conditions in microhabitats where vascular plants grow on particular relief microforms. This differs from previous research of microclimatological temperature changes conducted mainly in OTCs (Bokhorst et al. 2007; Bokhorst et al. 2008; Bokhorst et al. 2011; Bartak et al. 2019). However, some of those OTC studies also covered a relatively large area (Bokhorst et al. 2007; Bokhorst et al. 2008; Convey et al. 2018) and included measurements taken from environments that were not artificially modified (Walton 1982; Davey et al. 1992; Convey et al. 2018).
Analysis of the link between macro- and microclimate is crucial for understanding the transformation of meteorological fields during the macro-to-micro-scale transition. This study aimed to define the main features of LT variability and to analyse the relationship between LT (strongly affected by microclimate) and meteorological conditions in the areas where native vascular plants grow in the Argentine Islands–Kyiv Peninsula region. This paper discusses the following questions. How deterministic is intra-annual LT variability? What is the relationship between LT (representing micro-conditions) and other meteorological parameters measured at the weather station (representing the influence of meso- and macro-conditions)? Which meteorological parameters most strongly correlate with LT and are therefore helpful for monitoring terrestrial organisms, in particular vascular plants? What are the key meteorological parameters for the simple statistical approximation of daily LT in areas where terrestrial organisms exist?
Material and methods
Study area, instruments and measurement data
The area of study is part of Wilhelm Archipelago and Graham Coast. For the purposes of this study, we call our study area the Argentine Islands–Kyiv Peninsula region, which encompasses part of the coast of the Kyiv Peninsula and some of the islands off it, including Petermann Island, the Argentine Islands, the Yalour Islands, the Berthelot Islands and Darboux Island (Fig. 1, Supplementary Table S1).

This study is based on LT data measured in an area called the Bio-microclimatology Experimental Ground, which contains a set of points where HOBO Pendant Temperature/Light 64K Data Loggers collect data. The area is located mainly in the Argentine Islands, at a distance of 25 km around the Akademik Vernadsky Station (the meteorological station there corresponds to logger D1, number 31 in Fig. 1). The locations, short names and other information about the loggers are provided in Supplementary Table S1.
This study is focused on the micro-scale features of the polar climate that determine local flora growth. Over the course of a year, 37 mini-loggers were placed near populations of the native vascular plants Deschampsia antarctica and Colobanthus quitensis (Kunth Bartl.). It must be noted that the methodology of our LT measurements differs from the methodology of standard meteorological observations. The loggers were set on the surface (substrate) and as close to the plants as possible for the best representation of the thermal regime in which terrestrial surface organisms develop. The data measured by loggers cannot represent air, soil or plant leaf temperature, strictly speaking. Nevertheless, LT values are comparable to other standard temperature data because the same radiative and heat fluxes impact LT. These data are important for both meteorological and biological studies.
The installed loggers are waterproof, two-channel devices that combine temperature and light intensity measurements (Fig. 2). They are suitable for measuring soil, air and water temperatures. The accuracy of the instrument is ±0.5°C for positive temperatures and ±0.7°C for negative temperatures. They can be operated at a minimum temperature of -20°C. The response time is 10 min for air temperature and 5 min for water temperature measurements. The drift of the temperature sensor in the data loggers is less than 0.1°C/yr. So light intensity could be recorded, the loggers were not shielded from sunlight. The temporal resolution of the data is 30 min; however, daily average values were calculated for the purpose of this study.

The logger dies (Supplementary Table S1) was set near the plants that grow under the regular heating influence of diesel-powered electricity generators enclosed in a nearby hut. Most of the time, this logger measured positive LT. This logger is a subject of special interest due to warmer-than-average conditions.
Four sources of basic meteorological information, all collected at or within 200 m of the Akademik Vernadsky Station, were used in addition to the data from the mini-loggers: (1) daily observations of snow depth, precipitation and sunshine duration; (2) atmospheric air pressure, air and dew point temperatures, relative humidity at 2 m above ground, wind direction and wind speed at 10 m above ground measured using a Vaisala AWS-310 automatic weather station, with a temporal resolution of 1 min; (3) downward and reflected solar shortwave radiation fluxes, downward and upward longwave radiation fluxes, soil temperature at 1 cm depth and 2-m air temperature measured with a MicroClimate Monitoring Station (mainly developed by EMS Brno, and including sensors from various manufacturers), with a temporal resolution of 10 min.
Statistical methods
All the data for a particular day were averaged. To define the prevailing wind during a particular day, the daily mean wind direction was calculated using vector computations (Eqns. 1–3). First, the daily average u and v wind components were calculated based on N measurements of wind speed (V) and wind direction (D):

Second, the prevailing wind direction(D) was found using the following equation:
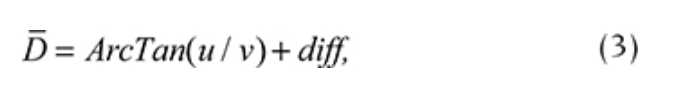
where diff = + 180 for ArcTan(u/v) < 180 and diff = -180 for ArcTan(u/v) > 180.
Wind speed was averaged as the arithmetic mean of V without using wind components.
The variance of LT is displayed using a box–whisker plot with median values, 25th and 75th percentiles and 5th and 95th percentiles for the non-outlier range. Outliers are beyond the 5th and 95th percentiles.
Intra-annual (seasonal) variations were estimated using harmonic analysis with the lowest frequency of one year. The highest frequency was limited to two months in order to defining the main variations at a macro-scale. Harmonic significance was estimated using a t-test with p = 0.05. Analysis of seasonality was based on mean values (LT1) calculated as the sum of significant harmonics (seasonal model):
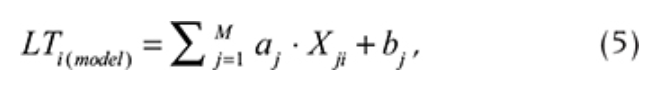
where n is the total number of k significant harmonics, LT is the mean value, Ak is the amplitude, ωk is the frequency (ωk = 2π/Tk, where Tk is the period of harmonic), φk is the phase and t is the time.
The dependence of LT on the values of the previous day was estimated using autoregressive models. A Pearson correlation was used for the analysis of the relationship between LT and meteorological parameters.
Statistical models based on multiple linear regression were used to estimate LT by other meteorological variables. The estimation was calculated using the following procedure. (1) Linear regression with every meteorological parameter was calculated, with LT acting as a dependent variable and meteorological parameter acting as an independent variable. (2) The most significant regression equation with the highest determination coefficient (R2) was found. (3) The significance of the equation was checked using a t-test. (4) If the equation was significant, LT was calculated using a single linear regression equation. (5) The obtained values from step 4 were subtracted from the input LT time series to find the residuals. (6) A new linear regression was calculated, where the residuals of LT were acting as the dependent variable, and every meteorological parameter was acting as an independent variable. Steps 2–6 were repeated until the regression was significant. As a result, an additive model was built for each time series of LT:
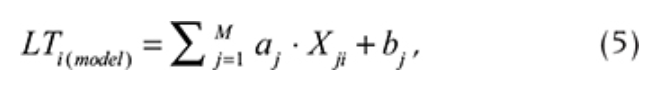
where LTi(model) is the modelled LT value for a certain day (i), Xji is the meteorological parameter (predictor), αj is the regression coefficient between LT and the meteorological parameter Xji, bj is the constant term (omitted if not significant) and M is the number of significant meteorological parameters (predictors).
We estimated the determination coefficient (R2) separately for seasonal variations of LT, autoregressive models and daily LT models using the following equation:

where MSS is the sum of squares of seasonal models/autoregressive models/daily LT models (variability explained) and TSS is the sum of squares of the time series (total variability).
Results
Intra-annual variability of LT
At a micro-scale, LT is a variable parameter; non-outlier ranges of the loggers’ time series vary from -11 to 12˚C (Fig. 3). The median temperature of all loggers varied from 0.0 to -3.0˚C (Fig. 3). Moreover, the medians for the majority of loggers are equidistant to the 25th and 75th percentiles, which indicates their closeness to mean values.
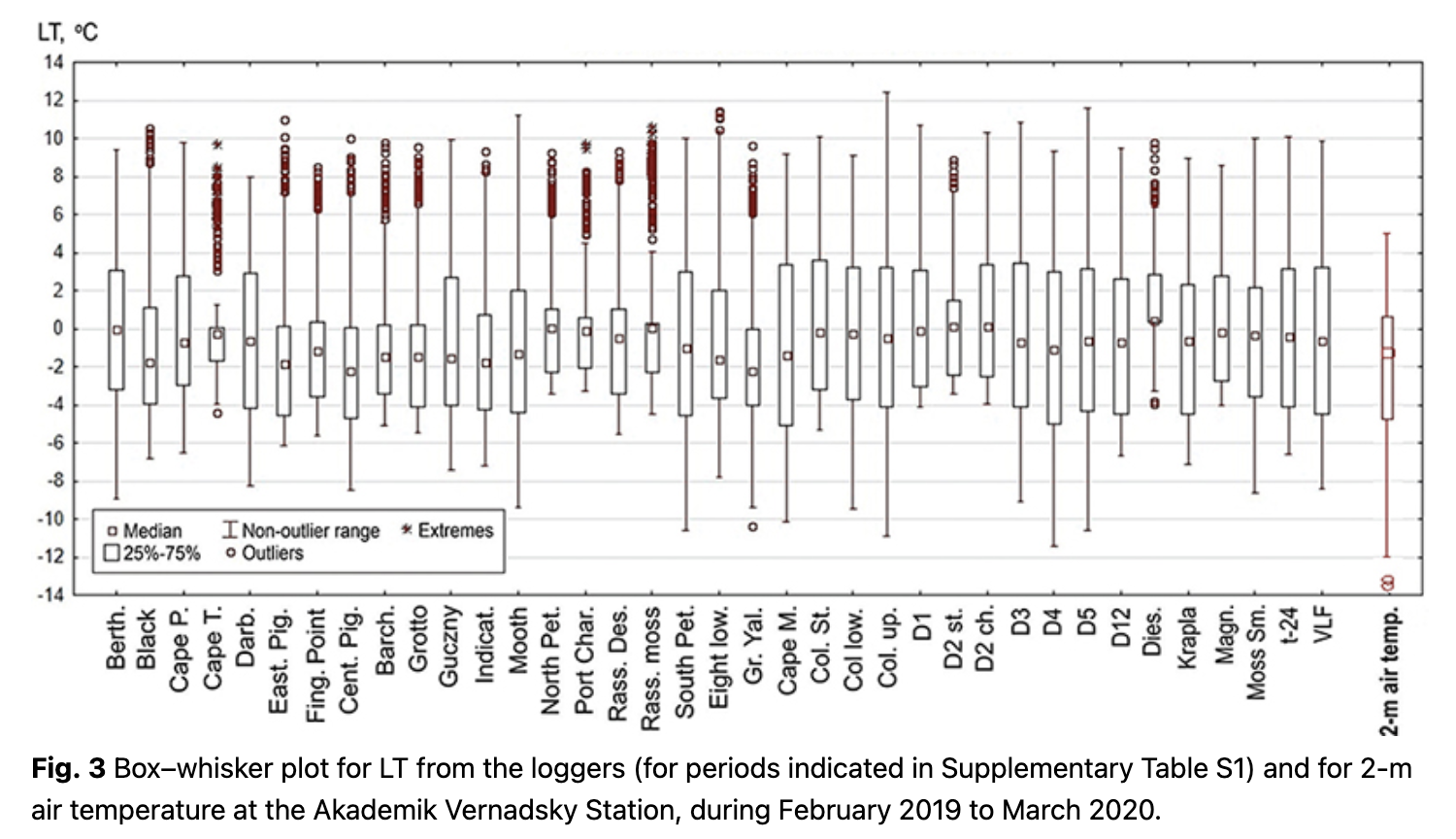
The lowest daily average LTs were between -10 and -12˚C, but for most locations, it was higher than -9˚C (see outliers and non-outlier range in Fig. 3). Overall, LT was higher than the 2-m air temperature measured at the Akademik Vernadsky Station. The highest daily LT varied from 8 to 13˚C, which was also usually higher than the daily 2-m air temperature at the station. There were more outliers for positive LTs.
It was found that LTs showed clear seasonality in all geographic locations. Determination coefficients (R2) of intra-annual variations varied from 0.75 to 0.93 (Fig. 4, Supplementary Table S2). This demonstrates that at least 75% of LT behaviour could be accounted for by periodical seasonal variations. Annual (first harmonic) and semi-annual (second harmonic) variations had the highest intensity and statistical significance at all locations.
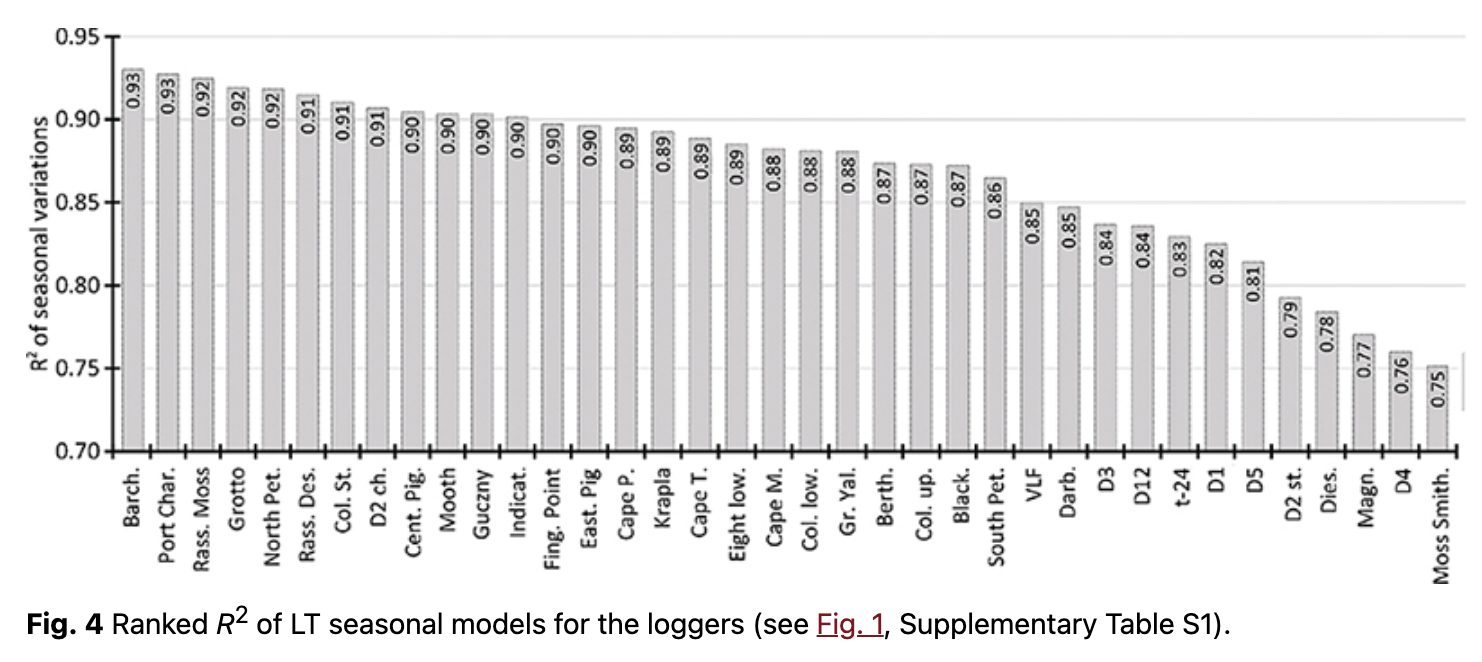
Our analysis shows that LT had distinct annual variations (first harmonic), which appeared as a result of temperature response to annual changes in incoming solar radiation at a given geographical location. The amplitude varied from 2.3 to 6.5˚C (Supplementary Table S2). Semi-annual variations had the second highest impact on LT intra-annual variability. The amplitudes of semi-annual variations varied from 0.4 to 1.8˚C, but it exceeded 1.0˚C for most loggers (Supplementary Table S2). The harmonic analysis demonstrated the significance of higher harmonics, but additional analysis is needed for more reliable conclusions. In general, clearer seasonality was observed on rock terraces, where the average R2 of seasonal variations was higher by 2–5%. The highest amplitudes of seasonal variations were observed on rock ledges. Moreover, the average first harmonic amplitude on open areas reached 5.2˚C, whereas it was ca. 4.6˚C in protected areas.
The annual cycle of LT calculated for each day, using Eqn. 4, is presented in Fig. 5. A stable 2-m air temperature transition through 0˚C was used to distinguish cold and warm periods. The differences in LT variability during the cold season were mostly responsible for the differences in R2 of the annual cycle. Changes in daily LT in January 2020 in the warm season were similar at all locations (Fig. 6a) and corresponded to the thermal regime formed by macro-scale climatic conditions over the region. Hence, LT variability during the warm period equally contributed to the R2 of annual variations for all loggers.
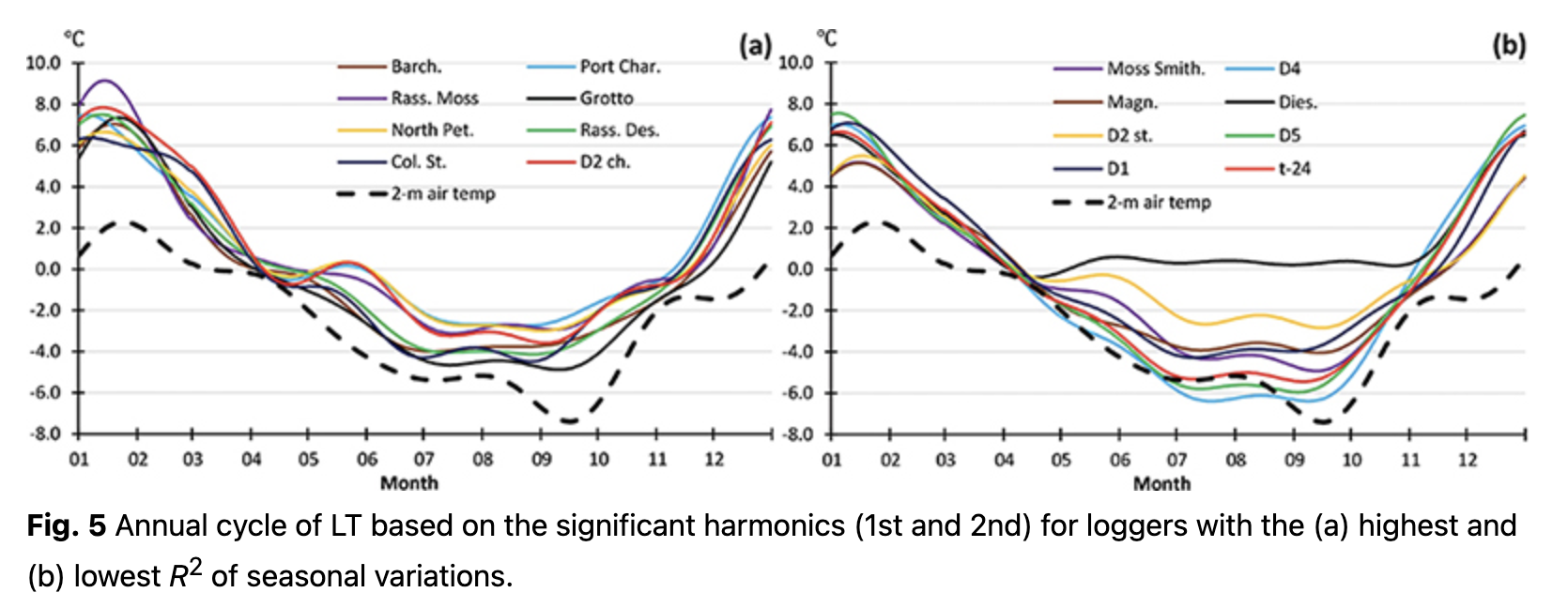
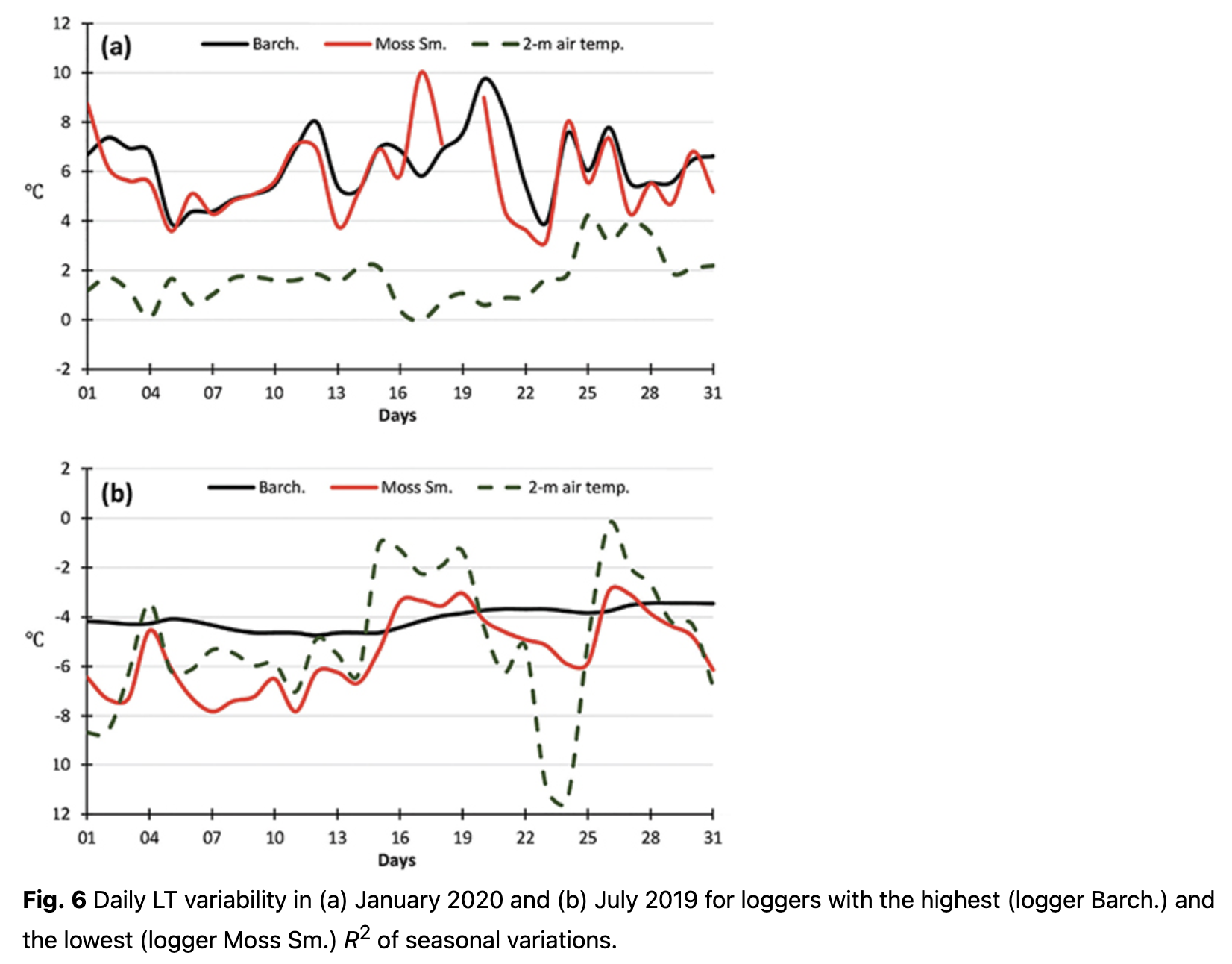
In contrast to the warm season, LTs varied significantly during the cold months and depended on the location (Fig. 6b). These differences impacted the significance (i.e., R2) of annual LT variability. Some locations had LT changes that were nearly consistent over the whole cold season with values of approximately -4˚C (e.g., logger Barch., Fig. 6b). These areas were most likely located under a thick layer of snow cover and could therefore not reflect air temperature changes over the region. As a result of such a steady LT value, annual variations for these loggers had higher R2. At other locations, LTs continued to replicate the air temperature regime with clearly visible day-to-day variability (e.g., logger Moss Sm., Fig. 6b). Higher day-to-day variability during the cold season resulted in lower R2 of the annual cycle.
After the seasonal variations were estimated, we calculated residuals, so any significant intra-annual variability was extracted from the time series. The residuals’ computations (seasonality removal) procedure was applied to each logger and each daily value (LTt) separately:
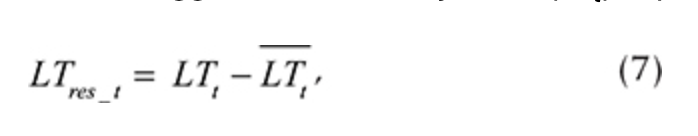
where LTres_t is the LT residual for each day t and LTt is the average value, computed using Eqn. 4. The autoregression of LT was analysed using residuals (LTres_t). It enabled us to determine the dependency on LT during the previous day (LTres_t-1), demonstrating how inertial LT was at a given location. R2 of the autoregressive part varied from 0.20 to 0.58 (Fig. 7, Supplementary Table S2).
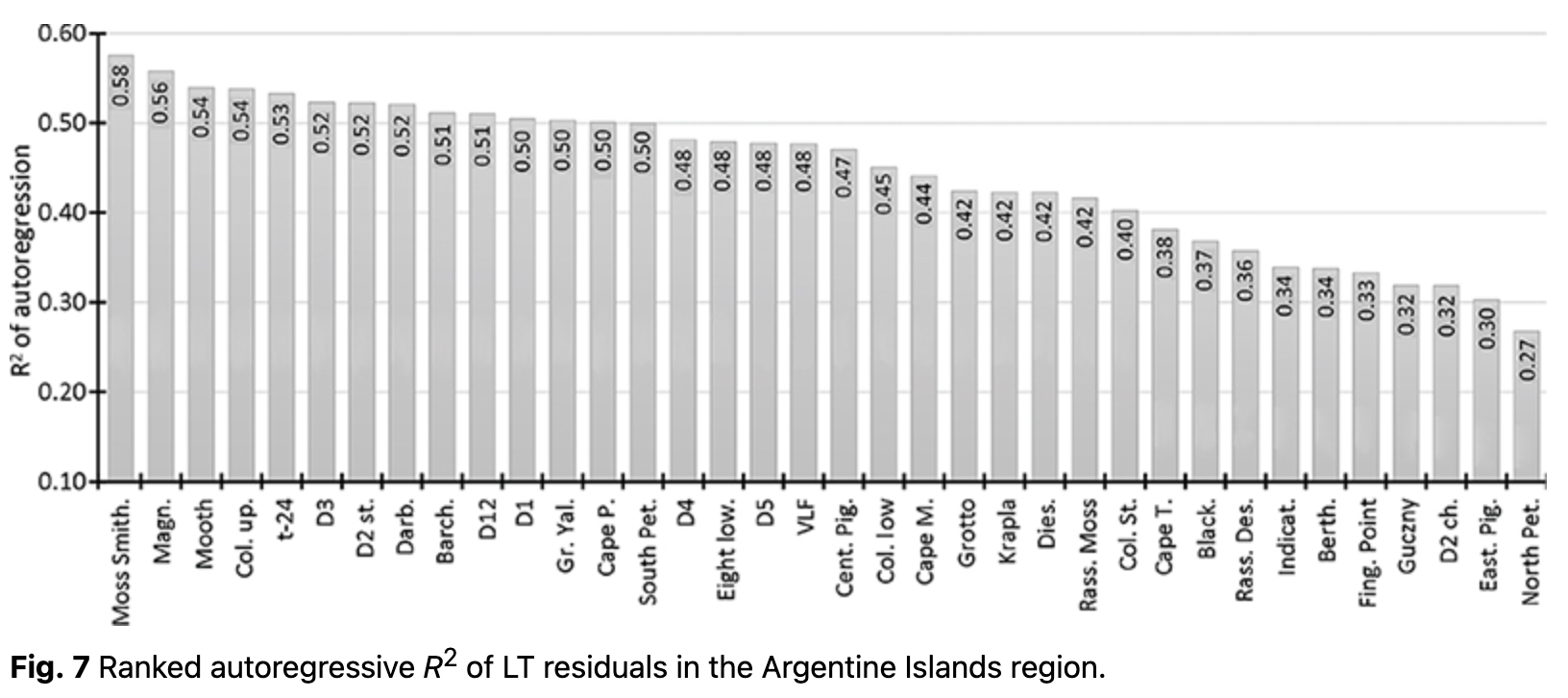
Loggers with higher R2 indicated locations where air temperature changes were slower, i.e., LT inertia was higher. In contrast, loggers with low autoregressive R2 values indicated more open areas where LTs were stronger, depending on external weather conditions. Typically, LTs on rock terraces and open areas showed lower R2 in autoregressive models, whereas the highest values were found on rock ledges, in clefts and in protected areas.
The sum of intra-annual variations and autoregressive parts gives a total R2 of 0.88–0.97. 3 to 12% of LT variability could not be described using the sum of periodic intra-annual changes and autoregression. The attempt to find dependencies between final residuals of LT and meteorological parameters did not show significant results. For some loggers, correlation was identified with moss surface temperature (r = 0.06…0.57) and air temperature (r = 0.11…0.60). The relationship between LT residuals and moss surface temperature could indicate that plants have a micro-scale influence on the thermal regime, in particular, areas where they grow, as shown by Hrbáček et al. (2020) and Perera-Castro et al. (2020). The dependence between LT residuals and air temperature is obvious: there are other processes with higher frequencies (e.g., the Southern Annular Mode, synoptic processes) that influence the air temperaWture and were not clearly indicated in LT variability.
Overall, our analysis has shown that the intra-annual variability of LT is rather deterministic, with less than 12% of unexplained variability. It indicates that the thermal regime in the areas where terrestrial organisms exist is not greatly isolated microclimatologically and closely reflects changes in the thermal regime at a macro-scale.
Using meteorological parameters to approximate LT
To demonstrate the strength of the relationship between LT and the macro-scale meteorological measurements representative for the region under study, we estimated the relationship between individual daily LT observations and meteorological variables from the Akademik Vernadsky weather station, the Vaisala AWS-310 automatic weather station and the MicroClimate Monitoring Station. As meteorological variables are interconnected, we analysed the relationships step by step from the highest correlation to the lowest one (which was still significant) using the procedure explained earlier.
As expected, correlations between LT and other temperature parameters were the strongest. The best correlations, ranging r = 0.63 to r = 0.81, were found between LT and 2-m air temperature (Fig. 8). Better relationships were found for loggers located on the rock slopes (e.g., Darb., Fig. 8), where LT fell below -5˚C during the period with negative air temperatures. This implies that these loggers were less insulated. The worst relationships between LT and 2-m air temperature were found on rock terraces (e.g., Port Char., Fig. 8) because the LT was quite stable during the cold period, with values of 0 to -4˚C, while 2-m air temperature frequently dropped below -10˚C. Overall, 2-m air temperature can be considered as the main predictor for LT at all locations, with regression coefficients varying from 0.336 to 1.035 (Supplementary Table S3).
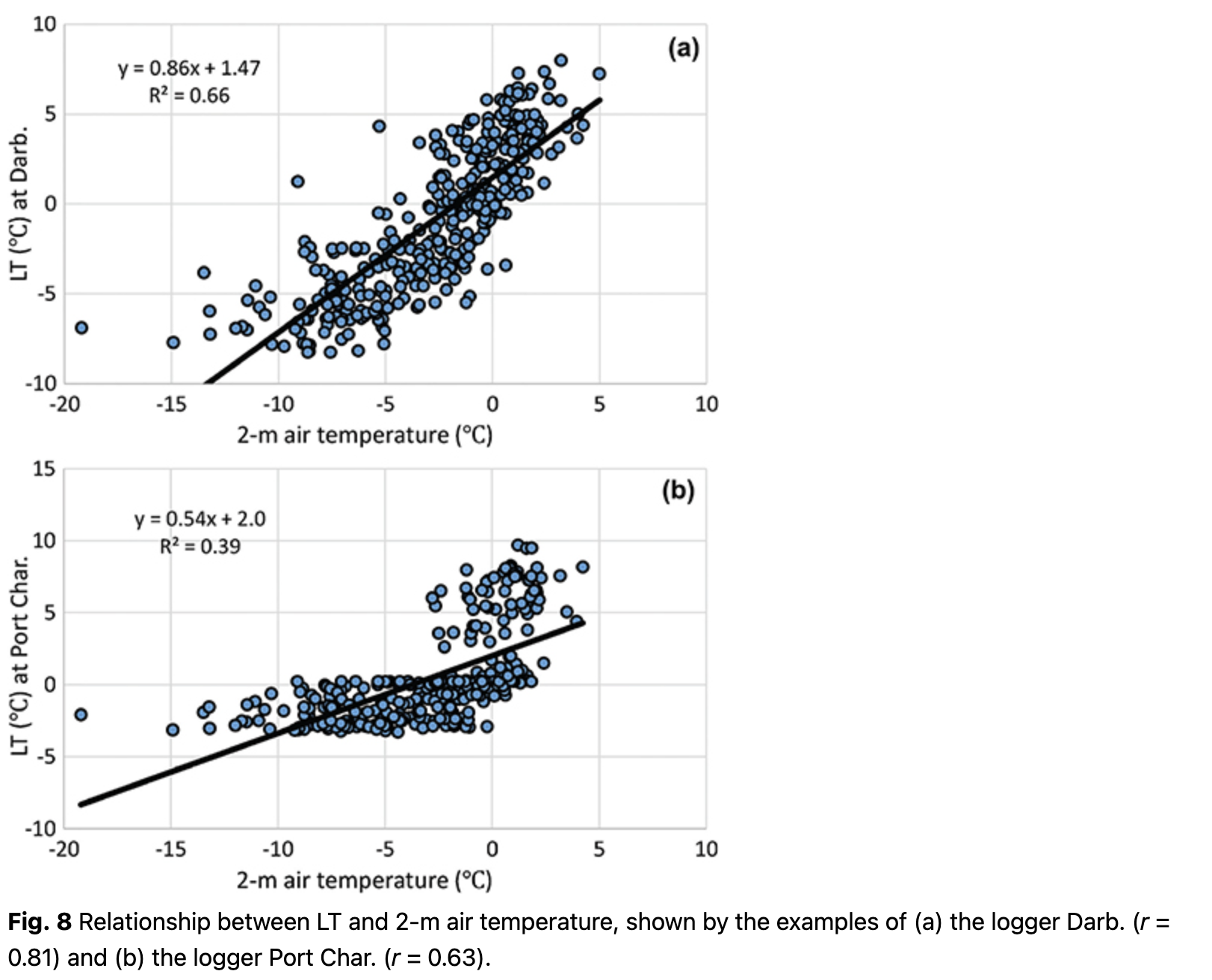
After the air temperature impact on LT was estimated, the regression dependences were subtracted from the time series. This procedure allowed us to avoid false significance with other parameters that correlate with air temperature.
The second step of calculations showed significance of wind speed. Correlations between LT residuals (with subtracted air temperature impact) and wind speed varied from r = 0.32 to r = 0.46 (Supplementary Table S3). The impact of wind speed was most pronounced on rock ledges. Regression coefficients were negative and ranged -0.329 to -0.616 (Supplementary Table S3); thus, as expected, greater wind speed increase caused 2-m and LT cooling.
At all locations, both 2-m air temperature and wind speed had the best relationships with LT. No other meteorological parameters were significant for all locations. They can therefore be used as key meteorological parameters for simple LT statistical modelling to estimate their joint impact. These models are the simplest way to combine LT at the micro-scale with meteorological parameters representing atmospheric conditions at a larger scale. As the regression models were calculated for every logger, their multi-model ensemble will help us to understand the LT conditions that allow plants to establish themselves in this harsh environment. The generalized equation for the daily average LT is:

where Tair is the daily averaged 2-m air temperature in ˚C, V is the daily averaged wind speed in m/s and a and b are regression coefficients (Supplementary Table S3).
Temporal distributions of observed LT with modelled values are presented in Fig. 9 for the cases of loggers with the highest and the lowest R2. Overall, R2 for the first model varies from 0.46 to 0.68 (Fig. 10). Some of the loggers, like Darb. (Figs. 9, 10), show a good fit between observed and modelled values even during the cold period. With negative air temperatures, these loggers have clear variations that could be modelled. Other loggers, like Port Char. (Figs. 9, 10), are characterized by an absence of variations during the cold period. During the cold period, low variance of observed LT (e.g., logger Port Char., Fig. 9) implied a thicker insulated layer over the logger, i.e., steady snow cover.
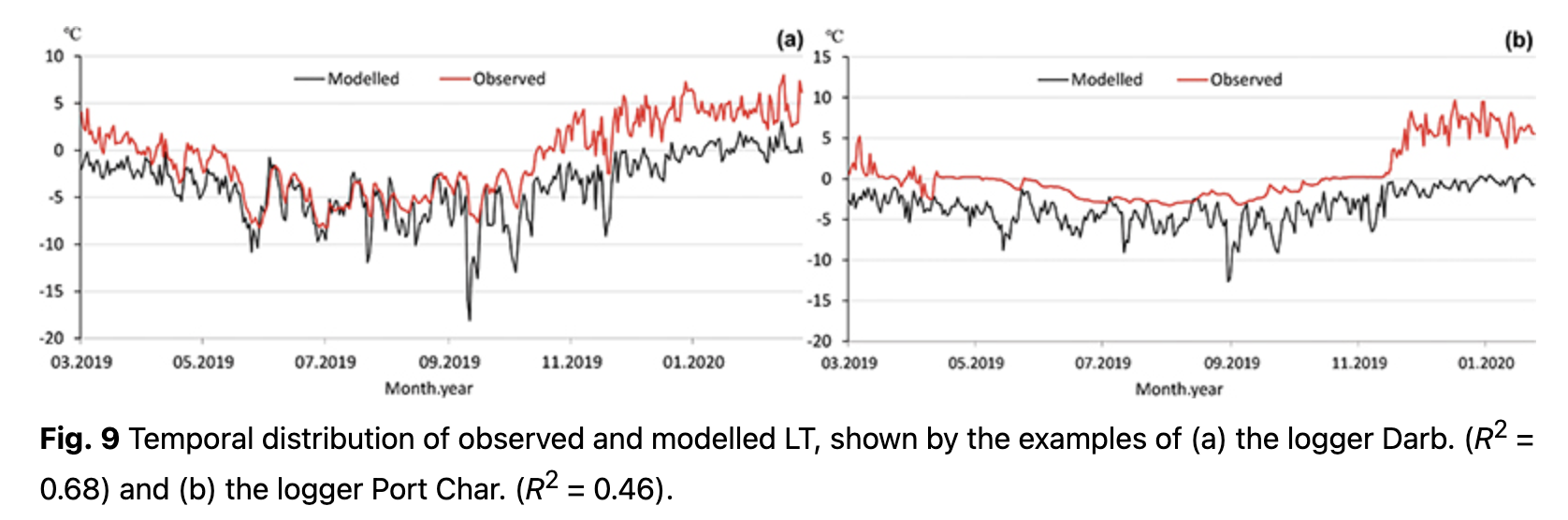
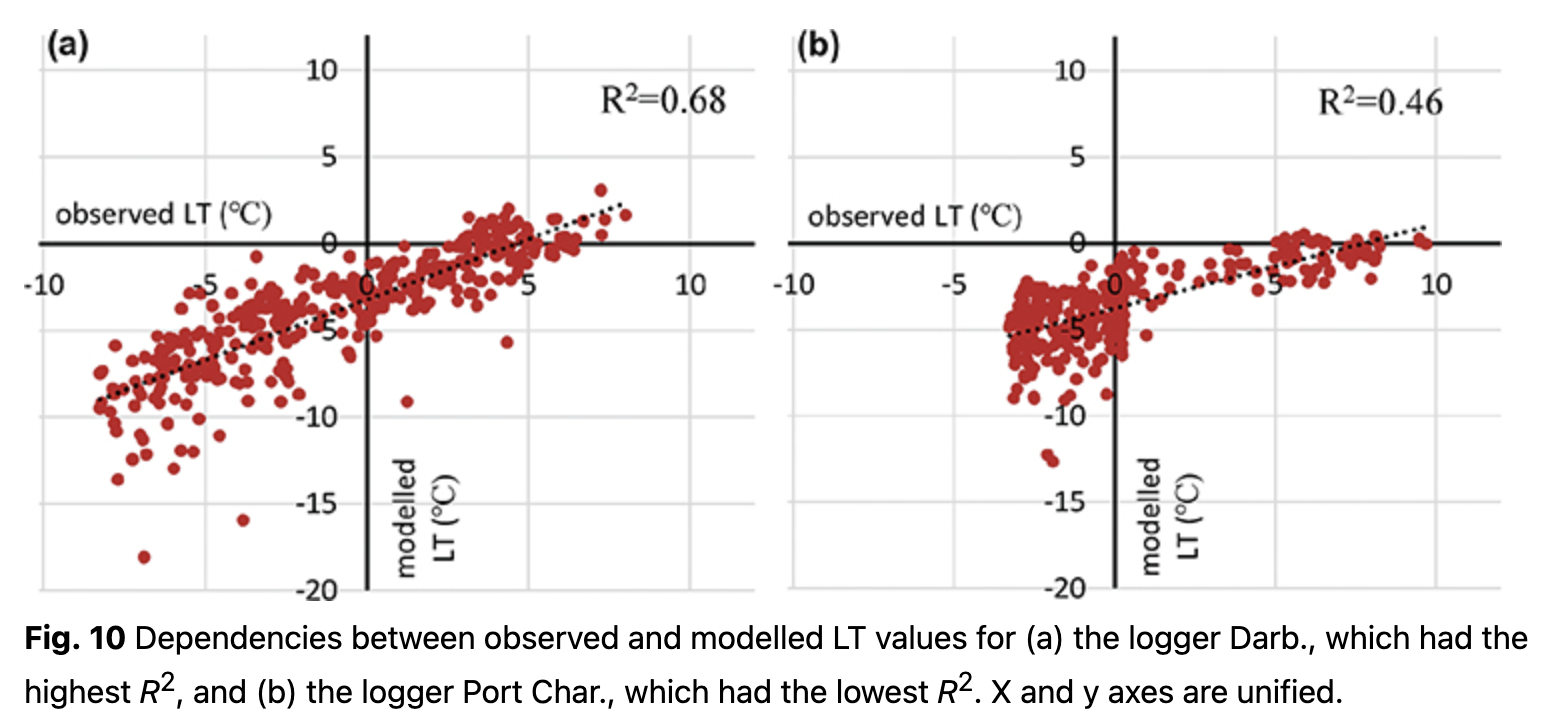
On the basis of the LT regression models, it is possible to indicate which loggers (and consequently which terrestrial organisms, in particular plants) were more open and less influenced by relief microforms. All the model values are cooler than the observed LT (Fig. 9). This indicates the role of other factors, some of which might relate to relief microforms and surface closeness.
Lower model R2 was more typical for rock terraces because of the lower regression coefficients of air temperature. In contrast, the best model results were for LT measurements on rock slopes, on the account of the better regression with air temperature, and at rock ledges because of closer relationships with wind speed. Dependency of model resulting from distance to the weather station and elevation was not found. No differences in model results were found between north and north-west expositions of the relief microforms on which all loggers were situated.
The statistical models based on regression dependencies from air temperature and wind speed with a set of calculated coefficients (Supplementary Table S3) could be used to build an ensemble to predict LT on various sites. In spite of the absence of direct measurements, temperature in the limited spots that allow plants to establish themselves could be approximated using these 37 regression equations.
It should be noted that the relationships between LT and air temperature at the station are formed under a variety of synoptic processes during the year. We analysed the impact of prevailing wind on temperature parameters for warm and cold periods separately. The relationship between LT and prevailing wind was similar for all loggers, but the daily average LT differed. West and south winds transported air masses from the ocean, and LTs reached higher values. Prevailing east winds brought air from the Antarctic Peninsula, and LTs were lower. This effect was especially strong during the cold period (Fig. 11b).
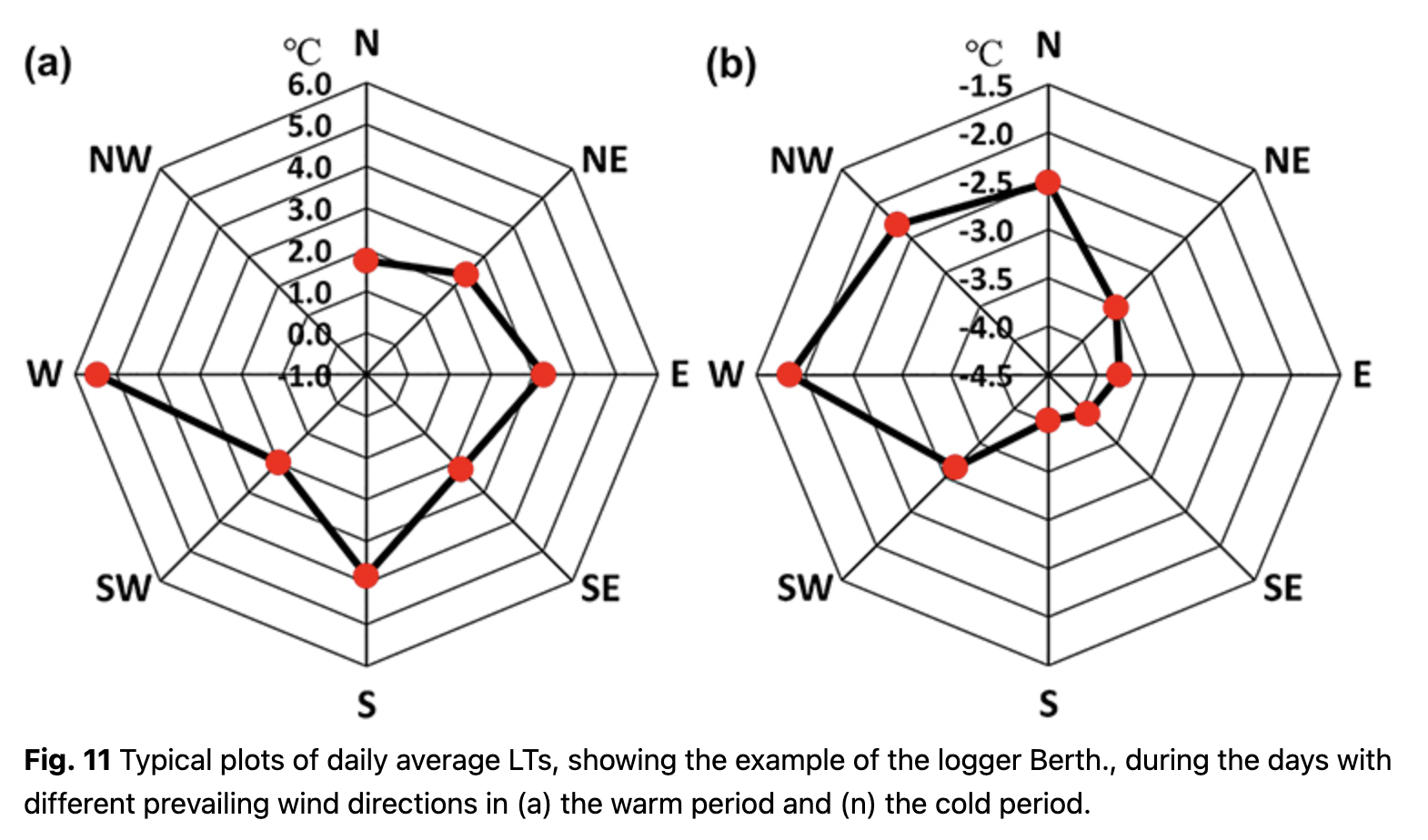
During the warm period, LT was less dependent on prevailing wind directions. Winds from the west brought the warmest air masses, although LT did not differ much between east and north winds. Statistically reliable data were absent for the north-west winds during the warm period (Fig. 11a) because only a few synoptic cases were observed.
No other meteorological parameters remained statistically significant for all locations once the impact of 2-m air temperature and wind speed was subtracted from the time series (Supplementary Table S4). Only sunshine duration and the upward longwave radiation flux remained statistically significant for loggers at some sites (Supplementary Table S4). The analysis did not reveal any clear relationships with daily precipitation sums, snow depth, downward longwave and downward and upward shortwave radiation fluxes. Although these parameters are crucial for the LT regime where plants grow, their measurements at a single site cannot be representative of other locations because of the heterogeneous nature of micro-relief forms and microclimates even within a fairly small area. After we considered air temperature and wind speed impact (using Eqn. 8) and calculated LT residuals, all other interconnected parameters lost their significance: soil and dew-point temperatures, relative humidity and air pressure (Supplementary Table S4).
Correlations between LT and sunshine duration varied from 0.04 to 0.48 (Supplementary Table S4). Coefficients were higher for north-facing rock ledges, whereas data from rock terraces did not show strong correlations. The higher correlations for many locations can be explained by certain land-cover types absorbing radiation.
LTs were correlated with upward longwave radiation flux at only 13 locations (Supplementary Table S4), with coefficients varying from -0.23 to 0.40. We did not find any similarity among locations where correlations between LTs and radiation fluxes were significant. Overall, the availability of radiation measurements only near the Akademik Vernadsky Station greatly limited the accuracy of the analysis.
Discussion and conclusions
We used experimental LT measurements that cannot strictly be considered as air, soil or plant leaf temperatures since the loggers were located on the substrate surface where native vascular plants grow. Nevertheless, our results have shown that LTs had typical seasonality and other variabilities of the temperature regime in the region and can represent the microclimatic conditions faced by native vascular plants at selected locations. Similar patterns were shown by Walton (1982), Guglielmin et al. (2012) and Convey et al. (2018), who demonstrated that air temperature influences the seasonal variability of ground temperature. Temperature seasonality has also been detected in the active ground layer (Guglielmin et al. 2012). In our study, LTs measured on the substrate surface showed clear seasonal variations, accounting for 75 to 93% of total LT variability, which is mainly caused by the significant annual and semi-annual variations. Better seasonality was observed on open rock terraces and lower on rock slopes and ledges. We have shown that differences in R2 of the LT annual cycle are formed mainly by the differences in day-to-day LT changes during the cold season (defined as a period with negative Celsius temperatures). Although our study was based on a short time series, the results are consistent with temperature variability in the Antarctic (see Davey et al. 1992; Van der Broeke 2000; Smith & Stammerjohn 2001; Turner et al. 2021). Overall, 3 to 12% of LT variability could not be described using the sum of periodic intra-annual changes and the autoregressive dependency.
On a daily scale, the highest correlation between LT (which represents microclimate conditions) and meteorological parameters (which represent the influence of meso- and macro-scale climatic conditions) was found with air temperature, with higher values at rock slopes compared to rock terraces and ledges. Wind speed was the second parameter that had significant correlations with LTs at all locations, with a better relationship on rock ledges. Both 2-m air temperature and wind speed could be used as the simplest way to estimate LT at a micro-scale in the vascular plants’ microhabitat, where no other regular and more complex measurements are possible. Better modelling results were obtained for locations where LTs had clear variability during the cold season under negative air temperatures. Worse results were for loggers with almost stable LT during the cold season. We were not able to measure snow depth at these remote locations with the loggers. However, our results showed the well-known effect of snow cover and its depth on the temperature regime close to the ground and on plants’ microhabitats (Davey et al. 1992; Bokhorst et al. 2016; Tarca et al. 2022). The almost constant LTs during the cold season corresponded to the locations with thicker insulating snow cover. Overall, LT could be estimated better at rock ledges (where stronger correlations with wind speed were obtained) and at rock slopes (where stronger correlations with air temperature were obtained). However, all the models simulated LT colder than observed.
Our study has confirmed that microclimatological differences of LTs appeared under the influence of incoming solar irradiance, which is extremely important for biota (Walton 1982; Bokhorst et al. 2012; Convey et al. 2018; Hrbáček et al. 2020). However, using radiation parameters for LT approximation presented a number of difficulties: the only available data were from distant meteorological stations; there was a possible warming effect for loggers exposed to direct solar radiation; cloudiness had a heterogeneous influence.
This present study helps us to understand LT variations at a micro-scale and how they correspond to macro-scale changes. Temperature conditions near the surface, where terrestrial organisms exist, have typical seasonal behaviour, but a few differences were revealed. Most remote locations made it impossible to provide regular meteorological measurements. Nonetheless, we have proposed a simple statistical approach based on the regression dependencies with 2-m air temperature and wind speed to estimate LT changes in the Argentine Islands–Kyiv Peninsula region. Because of the location of temperature loggers in native vascular plant microhabitat, it might also become possible to validate numerical weather models using temperature data from the most favourable areas for biological applications.
Acknowledgements
The authors acknowledge Kamil Laska and his work for allowing us to use data from the MicroClimate Monitoring Station, which has been supported by the Masaryk University project under MUNI/A/0952/2013 Analysis, Evaluation, and Visualization of Global Environmental Changes in the Landscape Sphere. The authors are grateful to Anna Evchun, at the State Institution National Antarctic Scientific Center, Kyiv, Ukraine, for preparing the map of the region and spatial distribution of loggers. The authors thank the reviewers whose valuable comments and constructive criticism allowed us to improve the paper. The authors thank David Crookall for help in manuscript proofreading. Last but not least, the authors acknowledge the Armed Forces of Ukraine, local Territory Defense Forces and volunteers for making it possible for us to complete this work.
References
| Abakumov E.V. & Parnikoza I.Y. 2015. Determination of the soil–permafrost border in two maritime Antarctic regions on the base of vertical electric sounding data. Ukrainian Antarctic Journal 14, 138–142, doi: 10.33275/1727-7485.14.2015.182. |
| Aerts R., Cornelissen J.H.C. & Dorrepaal E. 2006. Plant performance in a warmer world: general responses of plants from cold, northern biomes and the importance of winter and spring events. Plant Ecology 182, 65–77, doi: 10.1007/s11258-005-9031-1. |
| Bartak M., Laska K., Hajek J. & Vaczi P. 2019. Microclimate variability of Antarctic terrestrial ecosystems manipulated by open top chambers: comparison of selected austral summer seasons within a decade. Czech Polar Reports 9, 88–106, doi: 10.5817/CPR2019-1-8. |
| Bokhorst S., Huiskes A., Aerts R., Convey P., Cooper E.J., Dalen L., Erschbamer B., Gudmundsson J., Hofgaard A., Hollister R.D., Johnstone J., Jónsdóttir I.S., Lebouvier M., Van de Vijver B., Wahren C.-H. & Dorrepaal E. 2012. Variable temperature effects of open top chambers at polar and alpine sites explained by irradiance and snow depth. Global Change Biology 19, 64–74, doi: 10.1111/gcb.12028. |
| Bokhorst S., Huiskes A., Convey P. & Aerts R. 2007. The effect of environmental change on vascular plant and cryptogam communities from the Falkland Islands and the maritime Antarctic. BMC Ecology 7, article no. 15, doi: 10.1186/1472-6785-7-15. |
| Bokhorst S., Huiskes A., Convey P., Sinclair B.J., Lebouvier M., Van de Vijver B. & Wall D.H. 2011. Microclimate impacts of passive warming methods in Antarctica: implications for climate change studies. Polar Biology 34, 1421–1435, doi: 10.1007/s00300-011-0997-y. |
| Bokhorst S., Huiskes A., Convey P., van Bodegom M. & Aerts R. 2008. Climate change effects on soil arthropod communities from the Falkland Islands and the maritime Antarctic. Soil Biology and Biochemistry 40, 1547–1556, doi: 10.1016/j.soilbio.2008.01.017. |
| Bokhorst S., Pedersen S.H., Brucker L., Anisimov O., Bjerke J.W., Brown R.D., Ehrich D., Essery R., Heilig A., Ingvander S., Johansson C., Johansson M., Jónsdóttir I.S., Inga N., Luojus K., Macelloni G., Mariash H., McLennan D., Rosqvist G.N., Sato A., Savela H., Schneebeli M., Sokolov A., Sokratov S.A., Terzago S., Vikhamar-Schuler D., Williamson S., Qiu Y. & Callaghan T.V. 2016. Changing arctic snow cover: a review of recent developments and assessment of future needs for observations, modelling, and impacts. Ambio 45, 516–537, doi: 10.1007/s13280-016-0770-0. |
| Cannone N., Guglielmin M., Convey P., Worland M.R. & Favero Longo S.E. 2016. Vascular plant changes in extreme environments: effects of multiple drivers. Climatic Change 134, 651–665, doi: 10.1007/s10584-015-1551-7. |
| Cannone N., Guglielmin M., Malfasi F., Hubberten H.W. & Wagner D. 2021. Rapid soil and vegetation changes at regional scale in continental Antarctica. Geoderma 394, 115017, doi: 10.1016/j.geoderma.2021.115017. |
| Casanova-Katny M.A., Palfner G., Torres-Mellado G. & Cavieres L.A. 2014. Do Antarctic lichens modify microclimate and facilitate vascular plants in the maritime Antarctica? A comment to Molina-Montenegro et al. (2014). Journal of Vegetation Science 25, 601–605, doi: 10.1111/jvs.12122. |
| Convey P. 2011. Antarctic terrestrial biodiversity in a changing world. Polar Biology 34, 1629–1641, doi: 10.1007/s00300-011-1068-0. |
| Convey P. 2012. Polar terrestrial environments. In E. Bell (ed.): Life at extremes: environments, organisms and strategies for survival. Pp. 81–102. Wallingford: CAB International. |
| Convey P., Coulson S.J., Worland M.R. & Sjöblom A. 2018. The importance of understanding annual and shorter-term temperature patterns and variation in the surface levels of polar soils for terrestrial biota. Polar Biology 41, 1587–1605, doi: 10.1007/s00300-018-2299-0. |
| Convey P. & Smith R. 2006. Responses of terrestrial Antarctic ecosystems to climate change. Plant Ecology 182, 1–10, doi: 10.1007/s11258-005-9022-2. |
| Davey M., Pickup J. & Block W. 1992. Temperature variation and its biological significance in fellfield habitats on a maritime Antarctic Island. Antarctic Science 4, 383–388, doi: 10.1017/S0954102092000567. |
| Gerighausen U., Brautigam K., Mustafa O. & Peter H.U. 2003. Expansion of vascular plants on an Antarctic island as a consequence of climate change? In A. Huiskes et al. (eds.): Antarctic biology in a global context. Pp. 79–83. Leiden: Backhuys Publishers. |
| Gjessing Y. & Øvstedal D.O. 1989. Microclimates and water budget of algae, lichens and a moss on some nunataks in Queen Maud Land. International Journal of Biometeorology 33, 272–281, doi: 10.1007/BF01051089. |
| Graae B.J., De Frenne P., Kolb A., Brunet J., Chabrerie O., Verheyen K., Pepin N., Heinken T., Zobel M., Shevtsova A., Nijs I. & Milbau A. 2012. On the use of weather data in ecological studies along altitudinal and latitudinal gradients. Oikos 121, 3–19, doi: 10.1111/j.1600-0706.2011.19694.x. |
| Guglielmin M., Worland M.R. & Cannone N. 2012. Spatial and temporal variability of ground surface temperature and active layer thickness at the margin of maritime Antarctica, Signy Island. Geomorphology 155–156, 20–33, doi: 10.1016/j.geomorph.2011.12.016. |
| Hogg I.D., Cary S.C., Convey P., Newsham K.K., O’Donnell A.G., Adams B.J., Aislabie J., Frati F., Stevens M.I. & Wallh D.H. 2006. Biotic interactions in Antarctic terrestrial ecosystems: are they a factor? Soil Biology & Biochemistry 38, 3035–3040, doi: 10.1016/j.soilbio.2006.04.026. |
| Hrbáček F., Cannone N., Kňažková M., Malfasi F., Convey P. & Guglielmin M. 2020. Effect of climate and moss vegetation on ground surface temperature and the active layer among different biogeographical regions in Antarctica. Catena 190, article no. 104562, doi: 10.1016/j.catena.2020.104562. |
| Miotke F.-D. 1988. Microclimate, weathering processes and salt within ice-free continental Antarctica. Polarforschung 58, 201–209. |
| Molina-Montenegro M.A., Ricote-Martínez N., Muñoz-Ramírez C., Gómez-González S., Torres-Díaz C., Salgado-Luarte C. & Gianoli E. 2013. Positive interactions between the lichen Usnea antarctica (Parmeliacea) and the native flora in Maritime Antarctica. Journal of Vegetation Science 24, 463–472, doi: 10.1111/j.1654-1103.2012.01480.x. |
| Molina-Montenegro M.A., Torres-Diaz C. & Gianoli E. 2014. Antarctic macrolichen modifies microclimate and facilitates vascular plants in the maritime Antarctica—a reply to Casanova-Katny et al. (2014). Journal of Vegetation Science 25, 606–608, doi: 10.1111/jvs.12141. |
| Oliphant A.J., Hindmarsh R., Cullen N. & Lawson W. 2015. Microclimate and mass fluxes of debris-laden ice surfaces in Taylor Valley, Antarctica. Antarctic Science 27, 85–100, doi: 10.1017/S0954102014000534. |
| Parnikoza I.Y., Abakumov E.V., Dykyy I.V., Pilipenko D.V., Shvydun P.P., Kozeretska I.A. & Kunakh V.A. 2015. Vlijanie ptic na prostranstvennoe raspredelenie Deschampsia antarctica Desv. Ostrova Galindez (Argentinskie Ostrova, priberežnaja Antarktika). (Influence of birds on the spatial distribution of Desсhampsia antarctica [Desv.] on Galindez Island [Argentinean Islands, coastal Antarctic].) Vestnik of Saint Petersburg University, Series 3, Biology 1, 78–97. (In Russian with English abstract.) |
| Parnikoza I.Y., Abakumov E.V., Korsun S., Klymenko I., Netsyk M., Kudinova A. & Kozeretska I. 2016. Soils of the Argentine Islands, Antarctica: diversity and characteristics. Polarforschung 86, 83–96, doi: 10.2312/polarforschung.86.2.83. |
| Parnikoza I.Y., Berezkina A., Moiseyenko Y., Malanchuk V. & Kunakh V. 2018. Kompleksna harakterystyka rajonu Argentynskih Ostroviv ta Ostrova Galindez (morska Antarktyka) jak poligonu dlja vyvčennja dynamiki nazemnoji roslynnosti. (Complex survey of the Argentine Islands and Galindez Island [maritime Antarctic] as a research area for studying the dynamics of terrestrial vegetation.) Ukrainian Antarctic Journal 17, 73–101, doi: 10.33275/1727-7485.1(17).2018.34. (In Ukrainian with English abstract on the website.) |
| Parnikoza I.Y., Convey P., Dykyy I., Trokhymets V., Milinevsky G., Inozemtseva D. & Kozeretska I. 2009. Current status of the Antarctic herb tundra formation in the Central Argentine Islands. Global Change Biology 15, 1685–1693, doi: 10.1111/j.1365-2486.2009.01906.x. |
| Parnikoza I.Y., Miryuta N.Y., Ivanets V.Y. & Dykyi E.O. 2018. Vyznačennja zvedenogo latentnogo pokaznyka prystosovanosti (ZLPP) iz vrahyvannjam vnesku dejakyh pokaznykiv dovkillja dlja popyliacij Deschampsia antarctica Ostrova Galindez (morska Antarktika) v sezon 2017/2018. (Determination of the united quality latent index of adaptability [UQLIA] and contribution of some environmental parameters to it for Deschampsia antarctica populations, Galindez Island [maritime Antarctic] season 2017/2018.) The Bulletin of Vavilov Society of Geneticists and Breeders of Ukraine 16, 190–202, doi: 10.7124/visnyk.utgis.16.2.1057. (In Ukrainian with English abstract on the website.) |
| Perera-Castro A.V., Waterman M.J., Turnbull J.D., Ashcroft M.B., McKinley E., Watling J.R., Bramley-Alves J., Casanova-Katny A., Zuniga G., Flexas J. & Robinson S.A. 2020. It is hot in the sun: Antarctic mosses have high temperature optima for photosynthesis despite cold climate. Frontiers in Plant Science 11, article no. 1178, doi: 10.3389/fpls.2020.01178. |
| Saez P., Cavieres L.A., Galmés J., Gil‐Pelegrín E., Peguero‐Pina J.J., Sancho‐Knapik D., Vivas M., Sanhueza C., Ramírez C.F., Rivera B.K., Corcuera L.J. & Bravo L.A. 2018. In situ warming in the Antarctic: effects on growth and photosynthesis in Antarctic vascular plants. New Phytologist 218, 1406–1418, doi: 10.1111/nph.15124. |
| Savenets M., Pysarenko L. & Pishniak D. 2020. Microclimatic variations of land surface temperature on Galindez Island (western part of the Antarctic Peninsula). Ukrainian Antarctic Journal 2, 3–15, doi: 10.33275/1727-7485.2.2020.648. |
| Smith R. & Stammerjohn S. 2001. Variations of surface air temperature and sea-ice extent in the western Antarctic Peninsula region. Annals of Glaciology 33, 493–500, doi: 10.3189/172756401781818662. |
| Tarca G., Guglielmin G., Convey P., Worland M.R. & Cannone N. 2022. Small-scale spatial–temporal variability in snow cover and relationships with vegetation and climate in maritime Antarctica. Catena 208, article no. 105739, doi: 10.1016/j.catena.2021.105739. |
| Tin T., Fleming Z.L., Hughes K.A., Ainley D.G., Convey P., Moreno C.A., Pfeiffer S., Scott J. & Snape I. 2008. Impacts of local human activities on the Antarctic environment. Antarctic Science 21, 3–33, doi: 10.1017/S0954102009001722. |
| Turner J., Barrand N.E., Bracegirdle T.J., Convey P., Hodgson D.A., Jarvis M., Jenkins A., Marshall G., Meredith M.P., Roscoe H., Shanklin J., French J., Goosse H., Guglielmin M., Gutt J., Jacobs S., Kennicutt M.C., Masson-Delmotte V., Mayewski P., Navarro F., Robinson S., Scambos T., Sparrow M., Summerhayes C., Speer K. & Klepikov A. 2013. Antarctic climate change and the environment: an update. Polar Record 50, 237–259, doi: 10.1017/S0032247413000296. |
| Turner J., Colwell S.R., Marshall G.J., Lachlan-Cope T.A., Carleton A.M., Jones P.D., Lagun V., Reid P.A. & Iagovkina S. 2005. Antarctic climate change during the last 50 years. International Journal of Climatology 25, 279–294, doi: 10.1002/joc.1130. |
| Turner J., Lu H., King J., Marshall G.J., Phillips T., Bannister D. & Colwell S. 2021. Extreme temperatures in the Antarctic. Journal of Climate 34, 2653–2668, doi: 10.1175/JCLI-D-20-0538.1. |
| Van der Broeke M.R. 2000. The semiannual oscillation and Antarctic climate, part 5: impact on the annual temperature cycle as derived from NCEP/NCAR re-analysis. Climate Dynamics 16, 369–377, doi: 10.1007/s003820050334. |
| Walther G.-R., Post E., Convey P., Menzel A., Parmesan C., Beebee T., Fromentin J.-M., Hoegh-Guldberg O. & Bairlein F. 2002. Ecological responses to recent climate change. Nature 416, 389–395, doi: 10.1038/416389a. |
| Walton D.W.H. 1982. The Signy Island terrestrial reference sites: XV. Micro-climate monitoring, 1972–1974. British Antarctic Survey Bulletin 55, 111–126. |
| WMO 2010. Guide to the Global Observing System. 2010 edition. Updated in 2017. WMO-No. 488. Geneva: World Meteorological Organization. |





