Feasibility studies of the design of pharmaceutical development and industrial introduction of parenteral preparations on the basis of recombinant proteins


Published: Jan. 1, 2016
Latest article update: Sept. 20, 2022
Abstract
In the conditions of introduction of the proper practice of production of medicinal facilities on pharmaceutical enterprises in Ukraine, it is necessary to develop effective measures of projects management for creation of favourable conditions for modernisation of pharmaceutical production, innovative management and management of development projects. The optimization of pharmaceutical production of injection preparations, modernisation of the pharmaceutical industries, is being implemented in accordance with the standards of GM. Risks-control of pharmaceutical production of hi-tech parenteral preparations on a new level is carried out at the following stages: preparation of raw material, preparation of facilities and equipment; the process of preparation of solution (the stage of preparation of solution, the stage of sterile filtration; filling, small bottles, ampoules and cartridges; washing small bottles, ampoules and cartridges, preparation of materials for corking, sterilization of components; soldering of ampoules, rolling of small bottles and cartridges; sterilization and checking for impermeability). Decreasing the risk focuses on the processes of reduction and prevention of risk and its causes, when the risk exceeds the indicated acceptable level. Decreasing the risk can include the measures of declining of authenticity of harm. Conclusions regarding the measures related to decreasing the risk can bring to the exposure of potential threats and new hidden risks.
Keywords
Injection preparations, innovative management, management of development projects, good manufacturing practice, pharmaceutical production
In the conditions of introduction of the proper practice of production of medicinal products at pharmaceutical enterprises in Ukraine, it is necessary to develop effective measures of projects management for creation of favourable conditions for modernisation of pharmaceutical production, innovative management and management of development projects.
Optimization of pharmaceutical production of injection preparations and modernisation of injection areas is being carried out in accordance with the standards of GMP. Project management includes aseptic and sterile production, beginning from washing, sterilization of materials, by next aseptic production, completing eventual sterilization of pharmaceutical product, or sterile filtration, overflow and packing in aseptic terms, also plugs possibility of aseptic production of parenteral medical forms in vials and cartridges.
Over the past 10 years, the development of biotechnological methods allowed to create and implement new analogues of human insulin in curative practice, that possess additional therapeutic advantages at maintenance of valuable hypoglycemia activity. The examples of such introductions are analogues of insulin of urgent (LISPRO and ASPART) and prolonged (GLARGIN, DETEMIR) action.
The process of obtaining of insulin and its analogues in bacterial expression systems conventionally can be divided into the next stages: fermentation (growing of stamm-producent in a fermenter); separation of biomasses and disintegration of cellular suspension with the selection of bodies of including; selection and cleaning of recombinant proteins; enzymolysis of recombinant proteins; chromatography cleaning of insulin or its analogues; obtaining of crystalline insulin or its analogues.
Pharmaceutical preparations that are not accessible for thermal sterilization (sterilized by other methods: chemical, filtration, radiation), in particular bioorganic preparations, recombinant proteins are prepared in aseptic condition with further use of innovative methodologies: chromatography division, application of the special chromatography columns. The newest achievements of improvement of technological processes of receipt of parenteral preparations are considered in scientific reviews [1; 2].
Analysing the results of Aseptic Live Show industrial production of parenteral medicinal products of pharmaceutical industry enterprises specialized in the production of automatic and aseptic packing machines, one should note that the situation has radically changed. It is predefined by contradictions between principles of the old and new pharmaceutical engineering. The new pharmaceutical engineering needs some new technological and non-standard decisions related to reduction of time of disinfection and drying of capacities for parenteral medicinal products.
Sterility means the absence of all viable microorganisms including viruses. At present, a sterility assurance level (SAL) of 10–6 is generally accepted for pharmacopoeial sterilization procedures, i.e., the probability of not more than one viable microorganism in an amount of one million sterilised items of the final product. By extrapolating the reduction rates following extreme artificial initial contamination, the theoretical overall performance of the procedure of at least 12 lg increments (overkill conditions) is demanded to verify an SAL of 10–6. By comparison, other recommendations for thermal sterilization procedures demand only evidence that the difference between the initial contamination and the number of test organisms at the end of the process amount to more than six orders of magnitude. However, the practical proof of the required level of sterility assurance of 10–6 is not possible. Moreover, the attainability of this condition is fundamentally dubious, at least in non-thermal procedures. Thus, the question is discussed whether the undifferentiated adherence to the concept of sterility assurance on the basis of a single SAL of 10–6 corresponds with the safety requirements in terms of patient or user safety, costs and energy efficiency. Therefore, in terms of practical considerations, a concept of tiered SALs is recommended, analogous to the comparable and well-established categorization into “High-level disinfection”, “Intermediate-level disinfection” and “Low-level disinfection”. The determination of such tiered SALs is geared both to the intended application of the sterilized goods, as well as to the characteristics of the products and the corresponding treatment options [ 3; 4; 5; 6].
In the case of aseptic preparation, filling and production procedures, a mean contamination probability of 10–3 is assumed. In automated processes, lower contamination rates can be realized. In the case of the production of re-usable medical devices, a reduction of at least 2 lg increments can be achieved through prior cleaning in validated cleaning and disinfecting devices. By chemical disinfection, a further reduction of ≥5 lg increments is achieved. In the case of sterilized surgical instruments, an additional concern is that they lie opened in the contaminated air for the duration of the operation, at least in conventionally ventilated operating theaters. Finally, the amount of pathogens necessary to cause an infection must be considered. By logical consideration of all aspects, it seems possible to partially reduce sterility assurance levels without any loss of safety. Proceeding from this, we would like to make the following suggestions for tiered SAL values, adjusted according to the respective sterilization task: SAL 10–6 for heat-resistant pharmaceutical preparations (parenterals), suggested term: “Pharmaceutical sterilization”; SAL 10–4 for heat-resistant medical devices, suggested term: “High-level sterilization”; SAL 10–3 for heat-sensitive re-usable medical devices, under the precondition of a validated cleaning efficacy of >4 lg increments, suggested term: “Low-level sterilization” [6].
Methods. Planning, design of technological process, imitation design taking into account provocative terms, analysis of the systems and estimation of risks of pharmaceutical production.
Results and discussions.
The features of processes and operations of pharmaceutical production of hi-tech parenteral preparations include planning, architectural and air-technical decisions for the clean facilities of productive area, that provide the class of cleanness (B, С, D), elements of asepsis.
Imitation of the process of production of insulins in cartridges with the use of growth media of TSB on an injection area №1 on one model series of production of insulins, on the example of preparation of Farmasuline Н solution for injections 100 МЕ/ml for 3 mls in a cartridge, takes into account the features of production of this pharmaceutical product. Validation test of aseptic process of filling of cartridges is based on realization of planning researches – the simulation of aseptic process with the use of the growth media (periodicity of 6 + 1 months), pre-arranged by protocol of 01-04-ААА- of MFTP - 2.0 on an injection area №1. The aim of validation tests is in receiving of convincing results, that in high degree will confirm the authenticity, that process of preparing, filtration, filling, capping of cartridges is provided by the necessary level of sterility of product in accordance with the requirements of ST-N Ministry of health care of Ukraine 42-4.0:2011 Medicinal products.GMP.
The algorithm of validation tests of the process of aseptic filling of cartridges includes testing of such stages: preparation of shop floors (01-04 - MFTP - 1); preparation of equipment (01-04 - MFTP - 2); preparation of raw material and materials (01-04 - MFTP - 3); preparation of solution (01-04 - MFTP - 4); filtration of coregent (01-04 - MFTP - 4.1); filtration of solution (01-04 - MFTP - 5); filling and capping of cartridges (01-04 - MFTP - 6); filling and rolling of cartridges, implementation of aseptic manipulations (01-04 - MFTP - 7); checking of cartridges for impermeability (01-04 - MFTP - 8); visual inspection and sorting of gap-filling cartridges (01-04 - MFTP - 9); the microbiological monitoring during a technological process (01-04 - MFTP - 10); incubation of gap-filling standards (01-04 - MFTP - 11); inspection of cartridges (01-04 - MFTP - 12); verification of heighting properties of nourishing media (01-04 - MFTP - 13).
Quality and risk management includes the systematic processes, intended for co-ordination, facilitation and perfection of scientific decisions in relation to risks. For realization of quality and risk management of the pharmaceutical production of hi-tech parenteral preparations on the new area constructed in accordance with introduction of the authorial project the "Universal integrated model of organization of pharmaceutical production of parenteral preparations"© there should be conducted the following: determination of the problem related to the risk, relevant suppositions identifying risk possibility; collection of information on a potential danger; setting of leader and determination of necessary resources. Draft chart of control of the pre-arranged results in accordance with made decision for the process of risk management is drafted below.
The risk-management of aseptic production comes true in accordance with the ISO standard [7].
The risk estimation includes authentication of dangers; analysis and determination of risk size, related to the dangers. The estimation of risks of quality is begun with determination of essence of problem of quality [7; 8].
Discipline in realization effective risk management of aseptic production is regulator documents WHO, FDA, EMEA [8; 9; 10].
Normative documentation for parenteral preparations on the area of production: apparatus chart (PID); analytical and normative documentation (AND); specification of raw material and critical auxiliary materials for preparations on the area of production "Production of liquid medicinal facilities in vials and cartridges" (S); Technical Conception of Project (URS).
The formalized estimation of risks is conducted in accordance with the following chart (picture 1) :
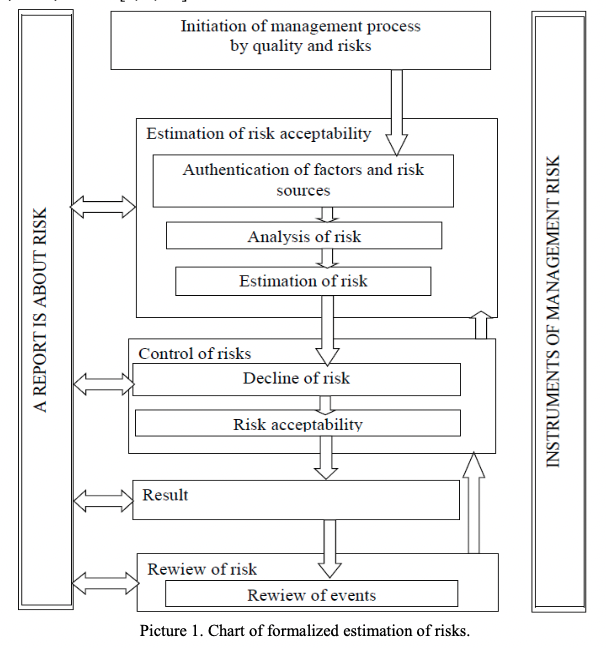
Risk control of pharmaceutical production of hi-tech parenteral preparations on a new area is performed on the following stages: preparation of raw material, preparation of areas and equipment; process of preparation of solution (stage of preparation of solution, stage of sterile filtration; filling in vials, ampoules and cartridges; washing of vials, ampoules and cartridges, preparation of materials for stoppering, sterilization of components; ampoule sealing, capping of vials and cartridges; sterilization and checking for impermeability).
Coming from mark reasons and degree of criticism of risks, methods of their minimization will be invented, so as methods of estimation of efficiency of the chosen measures. Therefore, exactly the estimation of risks allows working out the detailed plan of preparation to the test by nourishing media as a finishing chord of start of aseptic production.
Conclusion.
A perspective direction of researches of further development of the pharmaceutical industry and biotechnologies includes the development and industrial introduction of preparations of the prolonged action insulin based on the universal integrated model of cycle of pharmaceutical production of parenteral preparations according to ІСН.
Control of risk includes making decision with the aim of reducing to the acceptable level or acceptance of risk. Efforts given for risk control should be proportional to the risk value. Risk control determines the following: a risk which level is acceptable; what measures should be accepted for the declining of risk; what corresponding balance of risks and resources should be; whether there are new risks by the result of the controlled and identified risks of technological process.
Risk reduction focuses on the processes of reducing and preventing the emergence of risk, sources of origin, when the risk exceeds the rated acceptable level. Risk reduction may include measures of deviation of authenticity harm. The findings on the merits of the relative risk reduction could lead to the identification of potential threats and new hidden risks. Introduction of measures in relation to the risk reduction can lead to the exposure of potential threats and new hidden risks. Thus can be necessary to repeat estimation of risks for authentication of any changes of risk after application of risk reduction measures.






1. A Review on parenteral production technology / B. Venkateswara Reddy, B. Rasmitha Reddy, K. Navaneetha, V.Sampath Kumar // International Journal of Pharmacy and Biological Sciences ( IJPBS). 2013. Volume 3. Issue 1. P. 596-610.
2. Lippincott K. Williams. Remington, the Science & practice of pharmacy, Parenteral Preparation, 20th ed., volume‐I, Philadelphia: ISE publication; 2000. 804 p.
3. Bliem R, Nowak W.G. Inaktivierung von Mikroorganismen durch Sterilisation, Desinfektion oder Pasteurisierung. Pharm Ind. 2004. 66. Pp. 470–477.
4. Bliem R.F., Nowak G.W. Assessment of the Inaccuracy Inherent in the Exponential Model of Sterilization. Pharm Ind. 2004. 66. Pp. 780–785.
5. Decision trees for the selection of sterilization methods URL: (CPMP/ QWP/054/98http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guide line /2009/09/WC500003520.pdf (date of access: 20.05.2016).
6. Thomas von Woedtke, Axel Kramer. The limits of sterility assurance. Die Grenzen der Sterilisationssicherheit.// GMS Krankenhhyg Interdiszip. 2008. 3(3). Doc19. Published online 2008 Sep 3.
7. ISO 13408-1:1998 Aseptic processing of health care products – Part 1: General requirements.
8. Risk-MaPP: Manging the risk of cross contamination. Stephanie Wilkins at PharmaConsult US, Innovations in Pharmaceutical Techonology, EMA Statement on status of revision of Chapter 5 of the GMP guide concerning "dedicated facilities", London. 9 January 2008. Doc. Ref. EMEA/INS/GMP/14529/2008 (date of access: 20.05.2016)
9. FDA and Pharmaceutical Manufacturing Research Projects / Jeffrey T. Macher, Jackson A. Nickerson and all. URL: http://apps.olin.wustl.edu/faculty/nickerson/results (date of access: 20.05.2016).
10. WHO Expert Committee on Specifications for Pharmaceutical Preparations. Thirty-seventh Report. Geneva, World Health Organization, 2003. (WHO Technical Report Series № 908). Annex 7 Application of Hazard Analysis and Critical Control Point (HACCP) methodology to pharmaceuticals. P. 99 – 112.