Status and trends in the structure of Arctic benthic food webs


Published: May 20, 2015
Latest article update: Aug. 14, 2023
Abstract
Ongoing climate warming is causing a dramatic loss of sea ice in the Arctic Ocean, and it is projected that the Arctic Ocean will become seasonally ice-free by 2040. Many studies of local Arctic food webs now exist, and with this review paper we aim to synthesize these into a large-scale assessment of the current status of knowledge on the structure of various Arctic marine food webs and their response to climate change, and to sea-ice retreat in particular. Key drivers of ecosystem change and potential consequences for ecosystem functioning and Arctic marine food webs are identified along the sea-ice gradient, with special emphasis on the following regions: seasonally ice-free Barents and Chukchi seas, loose ice pack zone of the Polar Front and Marginal Ice Zone, and permanently sea-ice covered High Arctic. Finally, we identify knowledge gaps in different Arctic marine food webs and provide recommendations for future studies.
Keywords
Climate change, Arctic, trophic transfer, sea-ice retreat, pelagic-benthic coupling, food web
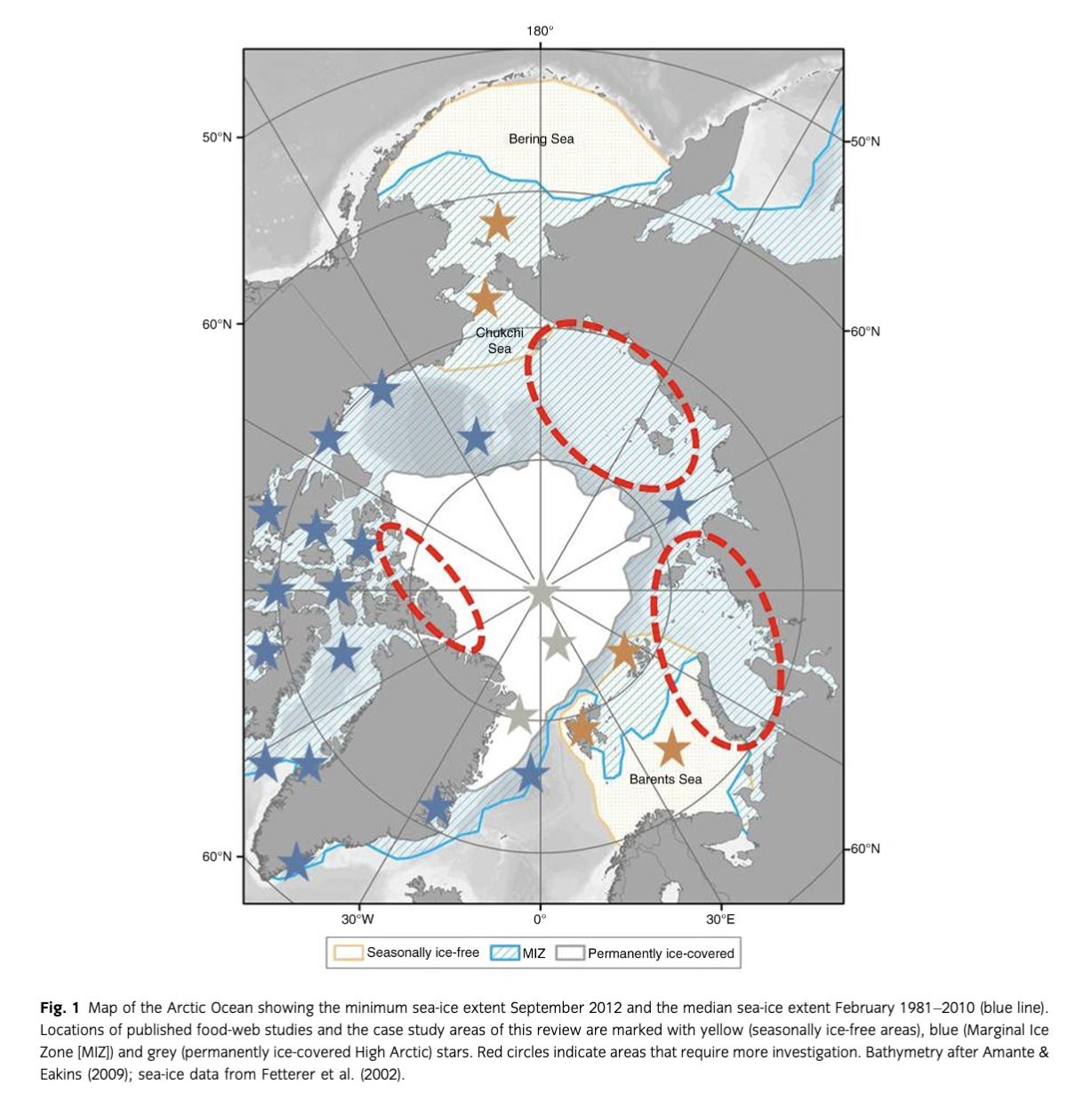
The Arctic Ocean is experiencing significant warming of approximately three times the global average (Steele et al. 2008; Serreze et al. 2009; Polyakov et al. 2010) and, despite a very pronounced seasonality, recent winter warming far exceeds that occurring in summer (Screen & Simmonds 2010). One of the characteristic features of the Arctic Ocean is sea ice, present permanently at high latitudes and seasonally at lower latitudes in winter (Fig. 1). The most conspicuous sign of warming is the dramatic loss of sea ice (Higgins & Cassano 2009; Parkinson & Comiso 2013): its summer extent has decreased by nearly 50% over the past decade (Fig. 1), and the Arctic Ocean has undergone a regime shift from multi-year ice to largely seasonal and much thinner ice cover (Comiso, 2012). The Arctic Ocean may become seasonally ice-free by as early as 2040 (Polyakov et al. 2010). Reductions in sea-ice cover are being further amplified by increased heat fluxes into the Arctic Ocean through the Bering (Woodgate et al. 2006; Woodgate et al. 2010; Woodgate et al. 2012) and Fram straits (Piechura & Walczowski 2009). This enhanced ocean temperature further delays the growth of sea ice in the fall (Steele et al. 2008). In terms of ecosystem functioning, these patterns indicate a shift towards an earlier spring transition between sea-ice-covered and sea-ice-free conditions (Grebmeier, Overland et al. 2006; Steele et al. 2008). Therefore, in the Arctic, climate change is not only affecting the physical structures such as sea ice, but is also responsible for multiple ecological changes on ecosystem functioning, including food-web structure, stability and efficiency, especially by affecting components at the base of the food web.
The two main sources of primary production in Arctic ecosystems are sea-ice algae and phytoplankton (Soreide et al. 2006). The growth of both ice algae and phytoplankton takes place within a one to four month period during spring and summer (Soreide et al. 2006; Renaud, Carroll et al. 2008; Iken et al. 2010). The productive season starts at the end of the polar night with sunlight triggering the bloom of sea-ice algae. Even though they constitute only a small-to-moderate portion ( <20%) of total annual primary production (Hegseth 1998; Gradinger 2009), ice algae contribute to Arctic benthic food webs during springtime on shallow continental shelves with seasonal ice cover because they occur early and sink fast following sea-ice retreat (Hobson et al. 1995; Tamelander et al. 2006). This ice algal contribution is proportionally more important in areas where sea-ice cover lasts later in the year (Gosselin et al. 1997) but is expected to decrease with current sea-ice retreat (Leu et al. 2011). A phytoplankton spring bloom follows the ice algae bloom as the sea-ice cover melts (Leu et al. 2011). In the summer, limited nutrients support a low phytoplankton biomass (Wassmann & Reigstad 2011), with episodic bloom conditions facilitated by occasional nutrient availability (Grebmeier, Cooper et al. 2006). These phytoplankton blooms, although responsible for the bulk of annual production, are usually restricted to open waters (but see Arrigo et al. 2012; Mundy et al. 2014). Boetius et al. (2013) also showed that sinking ice algae aggregates constitute an important food input to benthos in the Arctic basins. In addition to phytoplankton and ice algal production, microbial oceanic phototrophs can contribute up to 50% of total inorganic carbon assimilation and play a leading role in nutrient cycling (Falkowski et al. 1998). Bacterial primary production ratios have been estimated to reach more than 240% above 81°N (Rich et al. 1997) compared to 1-143% (32 + 6%) in the Barents Sea (Sturluson et al. 2008). The bacterial contribution to respiration in the water column may be substantial: 3-60% in the Chukchi Sea and Canada Basin, and 25% on average in the Arctic (Kirchman et al. 2009). Moreover, bacterial activity on sinking particles, an important habitat that harbours distinct communities of Arctic bacteria (Hansell & Ducklow 2003; Hodges et al. 2005), directly affects the quantity and quality of the organic matter that reaches the seafloor. In coastal areas and interior shelves, coastal erosion, river and glacial discharge can become important sources of terrestrial organic matter that can also be further utilized in benthic food webs (Dunton et al. 2006; Dunton et al. 2012; K^dra et al. 2012; Kulinski et al. 2014).
The quality and quantity of primary production, including ice algae and phytoplankton, reaching the seafloor have a strong impact on benthic communities (Pearson & Rosenberg 1978), which further cascades through the whole food web, especially in the highly seasonal Arctic marine ecosystems. Shallow Arctic shelves in particular are characterized by tight pelagic-benthic coupling due to low grazing in the water column during the bloom (Grebmeier et al. 1988; Grebmeier & McRoy 1989; Renaud, Carroll et al. 2008; Tamelander et al. 2008). This results in large export of organic matter, produced in the surface layers and descending to the seafloor and benthos (Grebmeier, Cooper et al. 2006), especially in spring, when production is far greater than zooplankton consumption (Tamelander et al. 2006). For example, almost 70% of the organic carbon produced in the water column during spring in the Pacific Arctic region reaches the seafloor (Walsh et al. 1989), supporting high biomass, abundance and diversity of benthic organisms (Grebmeier & McRoy 1989; Grebmeier, Cooper et al. 2006; Iken et al. 2010; Bluhm, Gradinger et al. 2011), which are important prey items for higher trophic level animals foraging on the seafloor, including diving sea ducks (Merginae), bearded seals (Erignathus barbatus), walrus (Odobenus rosmarus) and grey whales (Eschrichtius robustus) (Lowom et al. 2003; Grebmeier, Cooper et al. 2006; Grebmeier & Barry 2007; Grebmeier 2012). This contrasts with the summer period, when grazing of phytoplankton by zooplankton may reach up to 97% of daily water column primary production in more pelagic-oriented areas like the Barents Sea (Tamelander et al. 2006), thus limiting carbon export to the benthos. During summer, reworked organic matter consisting of zooplankton faecal pellets and carcasses, moults and bacteria, as well as phytodetritus, primarily fuels benthic food webs. Many shallow shelf Arctic systems, especially in the Pacific Arctic, have a high efficiency of energy transfer from the water column to the benthos (Ambrose & Renaud 1995). Macro- and megafauna appear to play a prominent role in carbon recycling (up to 30% efficiency for Arctic benthos; Clough et al. 2005; Renaud et al. 2007), but studies focusing on partitioning of carbon recycling for bacterial, meio- faunal, macrofauna and megafaunal components are rare (Piepenburg et al. 1995).
Trophic transfer efficiency describes the efficiency with which energy is transferred from one trophic level to the next, in particular the relative percentage of primary production that reaches top predators (Kozlovsky 1968). Species interactions, of which many are sea-ice related in Arctic ecosystems (Fortier et al. 2002; Gradinger & Bluhm 2004; Soreide et al. 2006; Gradinger 2009), control energy and organic matter flow which determine ecological efficiencies and can limit productivity and patterns of species dominance and food-web stability (McCann 2000). In the changing Arctic Ocean, species shifts and local extinctions and invasions may occur, leading to new interactions between species that have not co-evolved (Hobbs et al. 2006). New or missing links in established food webs may lead to large energy inefficiencies, changes in energy pathways that currently support key top predators, and destabilization of food-web dynamics (Vander Zanden et al. 1999; Pauly et al. 2002). However, this process is also dependent on the level of trophic and functional redundancy in a system (Layman et al. Understanding how Arctic food webs will become structured in the future will therefore entail disentangling the factors with most impact on interspecific interactions.
The aim of this paper is to present the current status of knowledge of the structure of various Arctic marine benthic food webs and their observed responses to ongoing climate change, in particular to sea-ice retreat. Potential changes to food-web trophic transfer efficiency are also discussed. To date, most focused studies of Arctic food webs are limited in space, time or taxonomic level (e.g., Iken et al. 2005; Aydin & Mueter 2007; Bergmann et al. 2009; Megrey & Aydin 2009; Iken et al. 2010; Feder et al. 2011; Dunton et al. 2012; Whitehouse et al. 2014), and large- scale syntheses across the Arctic are generally missing. Only a few pan-Arctic reviews have been published (Carmack & Wassmann 2006; Piepenburg et al. 2011; Wassmann et al. 2011), and none has focused on species interactions and trophic pathways of food webs nor on food-web structure and with trophic transfer efficiency. To complement existing studies, this review will focus on the key drivers of ecosystem changes in the Arctic, including sea-ice decline, temperature rise and changes in stratification and the consequences for important ecosystem functions that are susceptible to change, including nutrient regeneration, primary and secondary production, pelagic-benthic coupling, structure of food webs and consequences for top predators. In particular, we aim to: (I) identify key drivers of ecosystem change and potential consequences for ecosystem functioning; (2) present different scenarios and trends in Arctic marine food webs by analysing regional ecosystem case studies located along a sea-ice gradient: the seasonally ice-free Barents Sea and Chukchi Sea, the loose ice pack Polar Front (PF) and the Marginal Ice Zone (MIZ) and the permanent ice-covered High Arctic; and (3) identify knowledge gaps in Arctic marine food webs, and suggest possible methods to fill these gaps and provide recommendations for future studies.
Primary and secondary drivers of ecosystem change: potential consequences
Key drivers of ecosystem change
The primary driver of the observed ecosystem change in high latitudes is ongoing climate change, and warming in particular (Symon et al. 2005). Increasing air temperature is the main cause of sea-ice decline, along with the secondary drivers of Arctic ecosystem change, such as increase in sea-water temperature and altered stratification (Symon et al. 2005; Stocker et al. 2013). Sea ice is a major regulating component in controlling pelagic and benthic production through modulating water column stratification and light fields (Bluhm & Gradinger 2008; Gradinger 2009) because it controls the exchange of heat between the atmosphere and ocean and, together with snow cover, limits the penetration of light into the water column. Also, in the Arctic Ocean, the thaw-freeze cycle of sea ice and large freshwater riverine inputs result in pronounced haline stratification within the surface layer (Carmack & Wassmann 2006). Shortly after the phytoplankton spring bloom, the polar mixed layer becomes and remains nitrogen-depleted because of strong vertical stratification, which prevents replenishment during the summer season (Tremblay et al. 2008). Analysis of model data shows that when less sea ice is produced and freshwater load is increasing in the Arctic Ocean, the water column stratification becomes stronger, which decreases winter mixing (Slagstad et al. 2011), subsequently affecting nutrient distribution in the water column (11 et al. 2009; Codispoti et al. 2013; Matrai et al. 2013). In general, patterns of nutrients availability in the euphotic zone are a function of the total transport at the Barents Sea Opening and Fram Strait, through Davis Strait in the Canadian Archipelago and Bering Strait in the Pacific Arctic region, along with nutrient input from rivers, upwelling, stratification patterns and local mixing of deep water (Codispoti et al. 2013). Both major sources of nutrients in the Arctic Ocean, oceanic inputs associated with waters of Pacific and Atlantic origin, and riverine nutrient fluxes, are likely to change with accelerating climate change (Peterson et al. 2002; McClelland et al. 2006; Peterson et al. 2006; Shiklomanov & Lammers 2009). Changes in riverine freshwater runoff will likely be associated with changes in the quality and quantity of the nutrient supply, and while the loads of silicate, phosphate and dissolved and particulate organic matter are expected to increase in the future, trend in riverine nitrogen loads remain largely unknown (Frey et al. 2007; Raymond et al. 2007; Frey & McClelland 2009). Differences between nutrient transport and availability over the shelf areas and central Arctic may increase in the future: the already nutrient-depleted central Arctic Basin will likely become more oligotrophic, while shelf areas will be further enriched by increased transport of Atlantic and Pacific water masses and runoff. Biogeochemical processes and changes in adjacent basins as well as circulation changes may affect the future productivity of the Arctic Ocean.
Primary production and its consumers
Alterations of the seasonal cycle of primary productivity at the base of the food web is one of the most important consequences of rising temperature, sea-ice retreat and
changes in nutrient patterns. In general, ice algae and pelagic phytoplankton production occur sequentially during the year, with the abundance of ice algae relative to pelagic phytoplankton increasing northward coincident with greater sea-ice cover (Leu et al. 2011; Wassmann et al. 2011; Rubao et al. 2013). Although light availability and nutrients are often the prime limiting factors for primary production (Gradinger 2009; Leu et al. 2011), the timing of pelagic phytoplankton blooms is likely controlled by sea-ice retreat, whereas the timing of ice algae blooms is influenced by snow and sea-ice melt and starts earlier than open-water blooms. In many Arctic marginal seas, the timing of sea-ice retreat may have a strong impact on the timing of phytoplankton production, but little or no impact on the timing of ice algae peaks. Changes in the timing of maximum phytoplankton production influence the variability in time-lags between ice algal and phytoplankton production peak production (from 45 to 90 days; Ji et al. 2013). The timing of the seaice algal bloom is an important driver of spring secondary production as earlier ice algae bloom will export larger amounts of primary production to seafloor communities (Gradinger 1995), especially when water column grazing is low, strengthening pelagic-benthic coupling processes. Changes in timing of the pulses of ice algae and phytoplankton primary production and in the associated gap period between them will influence zooplankton grazer abundances and activities (Soreide et al. 2010; Varpe 2012). Although the timing of primary production varies greatly over time and space in different regions, systematic shifts in the timing of production and export processes have the potential to increase the frequency of mismatch between marine grazers and their food, which in turn can subsequently alter the organic matter flux to the seafloor and/or the transfer to the higher trophic levels (Rubao et al. 2013).
Because Arctic zooplankton reproduction is largely determined by the food quality of autotrophs (Muller- Navarra 2008), production of lipids in sea-ice diatoms early in the season is extremely important (Falk-Petersen et al. 2009; Soreide et al. 2010). These high-energy lipid compounds are rapidly transferred through the Arctic marine food chains. Changes in the initiation and duration of the growth season, and therefore in the timing, quality and quantity of sea-ice blooms, will directly affect, among others, calanoid copepods (Ji et al. 2012), which are critical to energy transfer between lower and higher trophic levels. Diatom lipids are accumulated in large amounts by Calanus species and ice amphipods, and further transferred to pelagic carnivorous zooplankton and pelagic fish stocks (Scott et al. 1999; Scott et al. 2001; Auel et al. 2002). Lipid levels increase from 10-20% in phytoplankton to 50-70% in herbivorous zooplankton and ice fauna that then become available as nutritious prey items for upper trophic level consumers (Daase et al. 2014). Dominant Arctic zooplankton taxa, like Calanus glacialis, switch from a diet of ice algae in spring towards phytoplankton in late summer, while others, like the sympagic amphipod Apherusa glacialis, feed mainly on ice algae during early spring (Falk-Petersen et al. 1999; Scott et al. 1999; Falk-Petersen et al. 2006). Ice-associated amphipods Onisimus spp. switch from ice algae in spring to an omnivorous diet by the end of the productive season (Werner & Auel 2005). The young ice amphipod Gammarus wilkitzkii feeds on ice algae, while their adults have a carnivorous diet, feeding preferentially on calanoid copepods (Scott et al. 2001; Werner et al. 2002). Although most benthic species do not feed directly on algal cells, changes in the timing and quality of bloom sedimentation (the latter additionally influenced by microbial processing) may impact those species' composition and abundances (Ambrose & Renaud 1997). Changes in the availability or abundance of ice algae or dependent zooplankton communities may cause cascading impacts on higher trophic level populations.
Microbial processes
Earlier sea-ice melt and increased stratification of the water column will promote picophytoplankton occurrence. Because of their short generation times, microorganisms respond quickly to environmental changes and are the first to react to global changes, affecting key ecosystem functions at the base of food webs (Sarmento et al. 2010). Significant changes in microbial communities and diversity have already been observed after a sharp decline of sea-ice cover in September 2007 (between 2002 and 2010; Comeau et al. 2011). Increased carbon supply from pelagic productivity and riverine discharge coupled with increased bottom water temperature could result in higher bacterial activity and rates of bacterial cycling of carbon (Kritzberg et al. 2010; Vaquer-Sunyer et al. 2010). The decreased quality and quantity of sinking particles (Wassmann & Reigstad 2011) and usable carbon (Renaud, Morata et al. 2008) being exported to the seafloor would decrease the efficiency of food webs. Picophytoplankton is believed to be relatively inaccessible as a prey for mesozooplankton (Li et al. 2009); however some studies suggest that protozoans may constitute a larger percentage of copepod diets (Campbell et al. 2009), even up to 80-90%, during summertime, when abundances, biomass and production are dominated by pico- and nanophytoplankton (Piwosz et al. 2009; De Laender et al. 2010; Piwosz et al. 2015) and when abundances of microzooplankton are high (Kubiszyn et al. 2014). In herbivore-limited systems, copepod production is therefore closely linked with protozoan production and constitutes a direct link between the microbial loop and higher trophic levels (Campbell et al. 2009; Sherr et al. 2009; De Laender et al. 2010; Nelson et al. 2014). Still, it is likely that Arctic food webs will lengthen at their base with the increase in numbers of trophic transfers within the microbial food webs or the number of grazers in the water column, decreasing the amount of carbon available for the higher trophic levels in the original food chain.
Our knowledge of the possible impact of viruses and parasitic protists in terms of their capacity to terminate algal blooms in the Arctic is limited (Nelson et al. 2014). Viral infection has been shown to control blooms of the coccolithophore Emiliania huxleyi in the Northern Atlantic (Wilson et al. 2002), while parasitic protists in the order Syndiniales have been coupled with collapses of blooms of dinoflagellate species in the Mediterranean Sea (Chambouvet et al. 2008). Increased impact of algal viruses and parasites would decrease the amount of food available for pelagic and benthic grazers. How these processes affect current blooms in the Arctic Ocean remains largely unknown, but both viruses and parasitic Syndiniales have been reported from sea ice (Wells & Deming 2006; 8achy et al. 2011; Collins & Deming 2011; Comeau et al. 2013; Piwosz et al. 2013) and the water column (Howard- Jones et al. 2002; Lovejoy et al. 2006; 8achy et al. 2011; Comeau et al. 2011; Payet & Suttle 2013).
Pelagic-benthic coupling
The initiation of sea-ice retreat is important for the timing, quality and amount of primary production (Springer et al. 1996; Hunt & Stabeno 2002), and changes in the timing of both ice algae and phytoplankton primary production may cause changes in food webs by influencing the standing stock of zooplankton, which will in turn affect the direct, ungrazed deposition of phytoplankton (Cooper et al. 2002) and subsequently benthic species (Overland & Stabeno 2004; Grebmeier, Cooper et al. 2006; Grebmeier, Overland et al. 2006; Nelson et al. 2009; Grebmeier 2012). In areas with reduced summer sea ice, pelagic grazing pressure will be higher (Lalande et al. 2007), perhaps altering sedimentation of organic matter, resulting in more pelagic-oriented systems. With increased primary production (Arrigo & van Dijken 2011; Arrigo et al. 2014; Palmer et al. 2014), pelagic food webs could become more productive and intercept more organic matter before it reaches the seafloor (Renaud, Carroll et al. 2008); however, nutrient limitation may halt this process. Increased grazing would reduce the export flux of carbon to the seafloor, weakening pelagic- benthic coupling processes. Studies in the Bering Sea found that with sea-ice cover the spring blooms were characterized by a higher proportion of diatoms, less recycling and greater export and, therefore, stronger pelagic-benthic coupling (Moran et al. 2012). In the case of open-water conditions, blooms were characterized by a higher proportion of dinoflagellates, greater carbon cycling in the water column and lower export to the seafloor, and consequently reduced pelagic-benthic coupling (Moran et al. 2012).
Upper trophic levels
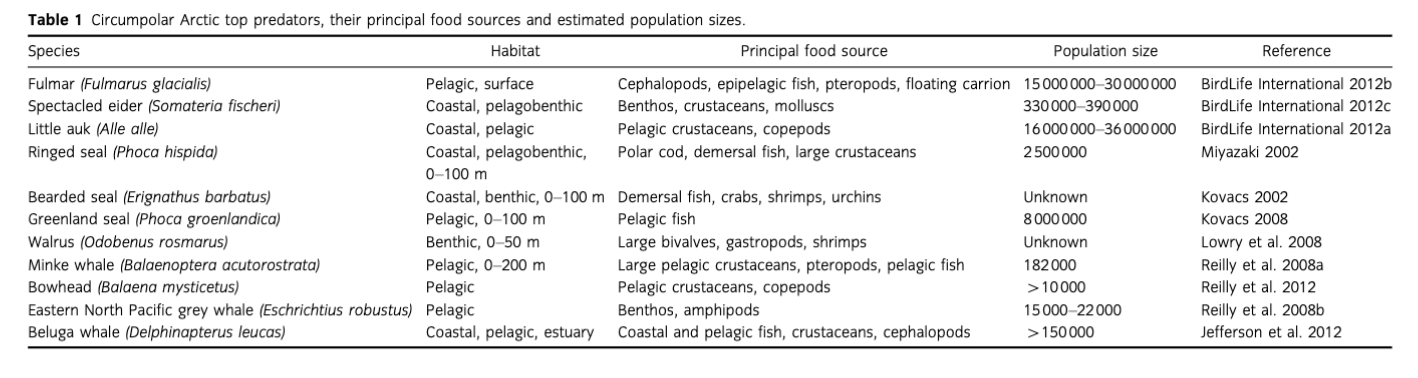
Changes in primary and secondary production affect upper trophic levels and are predicted to have increasing impact with climate warming (Wassmann 2006; Daufresne et al. 2009; Moore et al. 2014). Arctic marine top predators will have to face extreme changes in their habitat and forage base, including density and distributional shifts of their prey, as well as potential losses of some of their favoured lipid-rich prey species (Kovacs & Lydersen 2008; Kovacs et al. 2011). Upper trophic levels usually respond in a non-linear fashion to changes in ecosystem structure and usually the response depends on their exact position in the food web (Moore et al. 2014). Although the resilience of Arctic top predators is largely dependent on an individual's adaptive capacity, in general their resilience capacity depends largely on the region, the intensity and range of ice cover change and species characteristics (e.g., ice-obligate species are more vulnerable than ice-associated or seasonally migrant ones [Moore & Huntington 2008]). Sea-ice associated and seaice obligate species such as walruses and bearded seals that use sea ice as a platform for breeding, resting and foraging will be most affected by changes in sea-ice cover (Laidre et al. 2008; Moore & Huntington 2008; Table 1). Reductions in prey quality have been observed to have ecosystem-wide effects, such as population crashes of Steller sea lions (Eumetopias jubatus) in the Gulf of Alaska (Rosen & Trites 2000) and breeding failures of seabirds in the North Sea (Wanless et al. 2005). A decline in spawning and foraging areas will affect both predators and their prey. For instance, polar cod (Boreogadus saida) abundance might be significantly reduced, with tremendous consequences to the entire system as this species is believed to account for up to 75% of energy transfer between zooplankton and vertebrate predators (Damis et al. 2012). In addition to changes in resource availability, top predators will likely face increased complexity within food webs that tend to dissipate energy flow (W^slawski et al. 2009). This will likely be followed by increased competition from temperate species that are expanding northward, e.g., Atlantic cod (Gadus morhua), haddock (Melanogrammus aeglefinus) (Renaud et al. 2012) and Atlantic mackerel (Scomber scombrus) (Berge et al. 2015), and increased predation from species formerly unable to access them in areas of extensive sea-ice cover, such as killer whales (Orcinus orca) (Higdon & Ferguson 2009), gannets following northward move of herring and mackerel (Symon et al. 2005), and fish-eating whales that are getting more abundant in the Pacific Arctic region (Grebmeier 2012; Moore et al. 2014). Also, an increase in temperate and sub-Arctic fish migration is predicted to lead to a decrease in prey quality, since they are less lipid- rich than Arctic species (Symon et al. 2005; Hop & Gjosaeter 2013). Additionally, increased risks of disease and contaminants may also become an issue (Kovacs & Lydersen 2008).
Changes in species distribution ranges mediated by temperature
Temperature has a direct impact on metabolic and physiological processes as well as on the behaviour of individual organisms (Duarte 2007; O'Connor et al. 2007). It may influence growth, survival, reproduction, phenology and recruitment success of particular species (Lewis 1996; Walther et al. 2002; Lewis 2005; Herbert et al. 2007). Therefore, spatial distributions of organisms will likely change because of differential survival and recruitment of pelagic larval stages with varying water temperatures (Sirenko & Kolutin 1992; Blanchard et al. 2010; Grebmeier 2012). Changes in phenology can lead to a decoupling of the dynamics between predator and prey that will further alter current trophic relations and communities. Some species time their reproductive efforts to match the spring algal bloom (Falk-Petersen et al. 2009), e.g., females of the copepod Calanus glacialis utilize the spring pulse of ice algae to initiate reproduction, allowing their young to feed on the phytoplankton bloom that occurs after the breakup (Soreide et al. 2010). Along with temperature rise and sea-ice reduction, an increase in small-sized phytoplankton cells is predicted (Li et al. 2009) as well as a decrease in individual body size coupled with an increase in proportion of juveniles (Daufresne et al. 2009). At the population level, a shift of species may be observed, e.g., large, lipid-rich zooplankton species, such as C. glacialis and C. hyperboreus are being replaced by the smaller boreal and lipid-poorer species C. finmarchicus (Falk-Petersen et al. 2006).
Sea-water temperature rise and warmer Atlantic and Pacific waters adverted northward also represent a threat to Arctic biodiversity and may further change trophic relationships and food-web structure. These changes will facilitate open-water adapted species and boreal species to expand northward and ice-adapted species to retract in range (e.g., Sirenko & Gagaev 2007; Hollowed et al.) , which may lead to local extinctions, especially in the case of sea-ice dependent fauna (Clarke & Harris 2003). Changes have already been observed, including a northward distributional shift of fish and invertebrates in the Bering Sea (Mueter & Litzow 2008) and in the North Atlantic (Wienerroither et al. 2011), penetration of Pacific clams into the Chukchi Sea (Sirenko & Gagaev 2007), reoccurrence of Atlantic mussels in Svalbard (Berge et al. 2005), and Pacific zooplankton northward movement into the Beaufort Sea (Nelson et al. 2009). Also, an increase in year-round resident species may occur, e.g., grey whales that usually migrate south may stay in the Bering and Chukchi seas longer because of expanded open-water feeding areas and warmer water temperature (Moore & Huntington 2008).
Sea-ice gradient: scenarios and trends
Since sea-ice retreat is probably the most critical of expected consequences of climate warming for the Arctic marine ecosystems, in this review three regional ecosystem case studies are used to conceptualize possible changes in food-web structure and efficiency along a sea-ice gradient: the seasonally ice-free Barents Sea and Chukchi Sea, the loose ice pack PF and MIZ and the permanently ice-covered High Arctic (Table 2, Fig. 2a, b). In general, the described scenarios refer to current or possible future conditions on the shelves. The Barents and Chukchi seas were chosen as two case studies as
Table 2 Number of food-web studies undertaken in the Arctic by area. Data were collected using the search terms "Arctic" and "marine" and "food web" on the Web of Science. Studies that examined the relationship between two or more trophic levels were retained. Reviews and modelling with no in situ studies were excluded.
Area | Number of studies |
Barents Sea | 18 |
Bering Sea | 17 |
Beaufort Sea | 44 |
Canadian Arctic | 69 |
Central Arctic Ocean | 7 |
Chukchi Sea | 16 |
Greenland Sea | 23 |
Labrador Sea | 61 |
Laptev Sea | 1 |
Svalbard | 45 |
White Sea | 1 |
Yermak Plateau | 2 |
Total | 304 |
the food webs and associated trophic relations there are relatively well-studied (e.g., Iken et al. 2010; Feder et al. 2011; Renaud et al. 2011; Dunton et al. 2012; Grebmeier 2012; Kydra et al. 2012; Nelson et al. 2014; Table 2, Fig. 2). PF and MIZ are highly productive zones, which are likely to change their locations, extents and features as the ice edge retreats from the coast and continental shelves (Fig. 2). Arctic deep-sea regions represent a large part of the Arctic Ocean susceptible to change due to fast sea-ice retreat, yet studies of deep benthic food webs are scarce (Bergmann et al. 2009; Iken et al. 2010; van Oevelen et al. 2011; Table 2). hi all three case studies, the scale and the extent of response to climate change and sea-ice retreat remain largely unknown.
Arctic shelf: seasonally ice-free—Barents and Chukchi seas
Many Arctic shelf systems are characterized by high benthic biomass and production especially in areas of inflow of Atlantic or Pacific nutrient-rich water masses, and along the PF (Carmack & Wassmann 2006; Grebmeier, Cooper et al. 2006; Renaud, Morata et al. 2008; Fig. 2a). On the south-eastern Chukchi Sea shelf, primary production can exceed 430 g C m-2 y-1 (Springer et al. 1996; Sakshaug 2004; Lee et al. 2007); the Barents Sea has an estimated overall average annual primary productivity of about 100 g C m-2 y-1 and up to 300 g C m-2 y-1 in shallow banks (Sakshaug et al. 2009). Typically, about 44-67% of primary production in the Barents Sea reaches the seafloor (Wassmann, Reigstad et al. 2006; Wassmann, Slagstad et al. 2006) while it is up to 70% in the Chukchi Sea (Walsh et al. 1989). Unlike the Chukchi Sea, the Barents Sea supports immense fisheries and a high density of cetaceans. Pelagic foraging pisdvores and nesting seabirds are twice as abundant in the Barents Sea than in the Chukchi Sea (Hunt et al. 2013). In the Barents Sea, seasonally resident cetaceans are four to five times more abundant than in the Chukchi Sea. On the other hand, the density of pinnipeds and benthic-foraging whale species in the Chukchi is twice that of the Barents Sea, indicative of the Chukchi Sea being more of a benthic-driven system than the Barents Sea (Hunt et al. 2013). Indeed, Chukchi soft sediment infaunal and epifaunal communities are among the most productive in the world, reaching up to 50-100 g C m-2 or up to ca. 4 kg wet wt m-2 (Grebmeier et al. 1988; Grebmeier, Overland et al. 2006; Feder et al. 2007), while in the most productive areas of shallow banks in the Barents Sea the benthic fauna reaches up to 30 g C m-2 or 1.5 kg wet wt m-2 (K^dra et al. 2013).
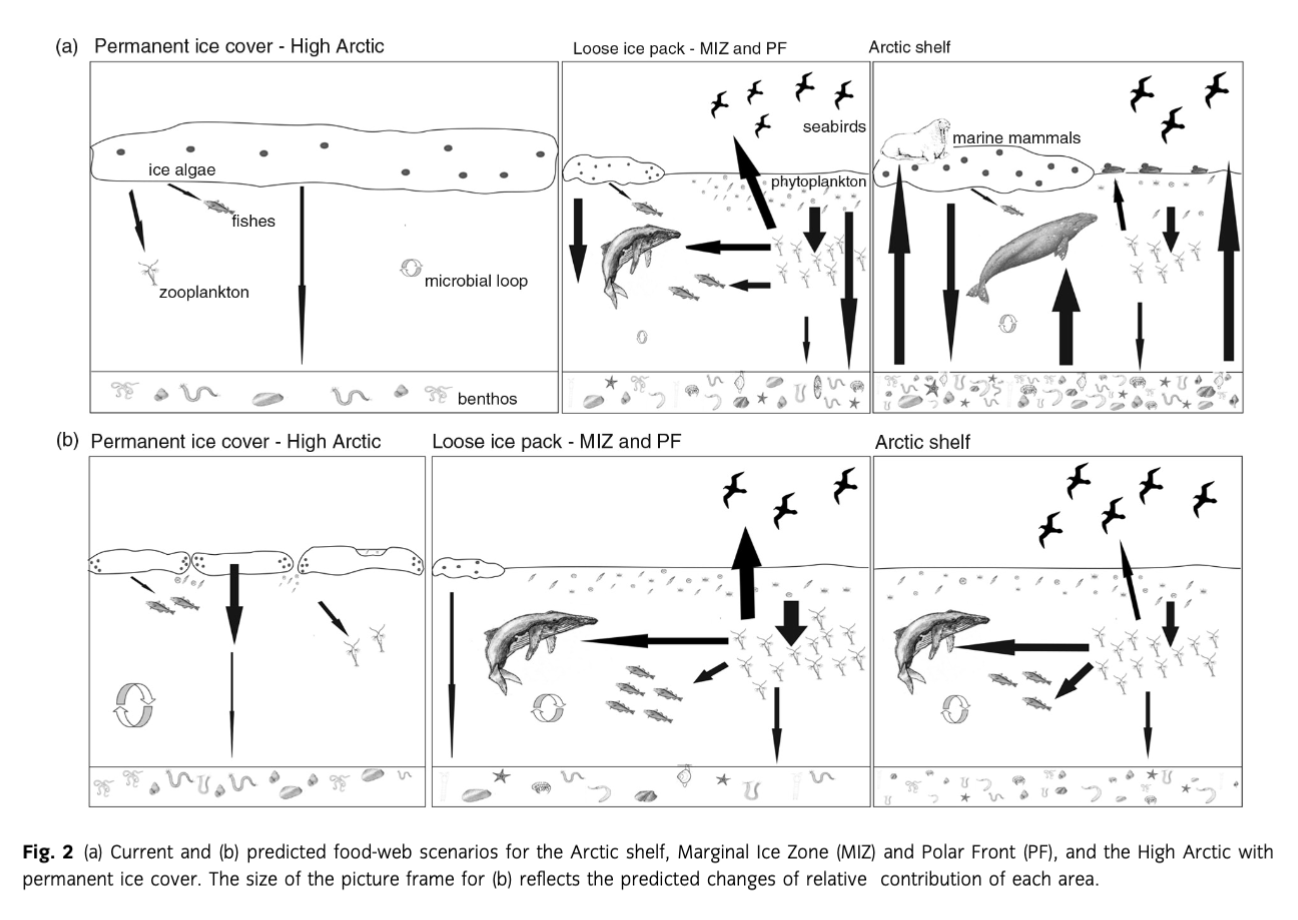
The ice edge is retreating northwards and, in the near future, ice-free summers and seasonal ice cover in winter may become typical for the shelf seas (Fig. 2b). Increases in primary production and phytoplankton biomass are predicted, as well as have been measured, as a consequence of sea-ice retreat and temperature rise (Gradinger 1995; Arrigo et al. 2008; Arrigo & van Dijken 2011; Wassmann & Reigstad 2011). Moreover, increased advection of Atlantic and Pacific waters into the Arctic Basin in the last decade (Walczowski & Piechura 2006; Woodgate et al. 2006; Piechura & Walczowski 2009; Woodgate et al. 2010; Walczowski et al. 2012) resulted in a 30% local increase of total primary productivity due to a greater proportion of smaller boreal planktonic species (Leu et al. 2011). Apart from quantitative changes in primary production, the quality and seasonality of primary production are expected to change because of the earlier onset of ice melt. Various stages of the reproductive cycle or increased activity of some Arctic benthic animals are timed to coincide with peak periods of organic matter deposition (Blake 1993; Renaud et al. 2007), so any change in seasonality, quantity or quality of food input may create a mismatch with faunal reproductive cycles (Renaud, Carroll et al. 2008). A shift from a benthic- oriented ecosystem with relatively low zooplankton stocks and strong pelagic-benthic coupling to a system dominated by pelagic food webs has already occurred in the northern Bering Sea in 1970s and 1980s (Overland & Stabeno 2004; Grebmeier, Overland et al. 2006) and benthic productivity has been decreasing over the past two decades in the northern Bering and southern Chukchi seas (Moore et al. 2003; Grebmeier, Overland et al. 2006; Grebmeier 2012).
Loose ice pack—MIZ and PF
A significant feature of the recent (2007-2012) decrease in ice extent has been the retreat of the ice edge away from the coast and continental shelves. One of the most obvious impacts has been the northward expansion and widening of the MIZ, a dynamic and biologically active band of sea-ice cover adjacent to the open ocean (Strong & Rigor 2013). MIZ width is a fundamental feature for polar ecosystem functioning and climate dynamics (Wadhams 2000). It is an interfacial region that forms at the boundary of open and frozen ocean and protects the stable morphology of the inner ice from wave penetration (Squire 2007). In the MIZ, interactions between sea-ice and the open sea result in modification of the properties of the ice compared to areas deeper within the pack (Weeks 2010). Significant forcing that impacts the sea ice in the MIZ results in varying surface roughness (Gupta et al. 2014), which affects prevalent physical and biological processes in the MIZ, such as wave dynamics (Wadhams et al. 1988; Squire et al. 1995), heat (Perovich et al. 1989), salt fluxes (McPhee et al.) and floe size distribution (Lu et al. 2008). It can also create potential habitats for organisms inhabiting the ocean-ice system (e.g., Arctic cod [Fortier et al. 2006]). Properties of the MIZ relative to neighbouring ice pack can markedly affect the carbon cycle and behaviour of microorganisms and top-level predators (Dunbar & Leventer 1987; Arrigo et al. 2012). The MIZ has been a long-standing feature in many Arctic shelf seas, like in the Bering, Chukchi and Barents seas, but is a relatively new phenomenon in higher latitude regions such as the deep Beaufort Sea and Canada Basin (Shimada et al. 2006). The MIZ is advancing poleward into regions where sea ice has become increasingly younger and thinner at the beginning of the annual melt (Strong & Rigor 2013). At the time of minimum sea-ice extent in September, the sea-ice edge is located over the deep Arctic Ocean, exposing large areas of previously permanently ice- covered waters, and MIZ-type conditions are becoming more prevalent in the High Arctic with the advancement of climate change.
Loss of sea ice will likely change the amount and characteristics of primary production in the MIZ (Bluhm & Gradinger 2008; Fig. 2b). Increased summer sea-ice melt will increase the overall extent of the MIZ over the shelves and lead to increased primary productivity (Anderson & Kaltin 2001). However, export fluxes largely depend on the coupling processes in the water column, including grazing, and therefore may not increase with warmer temperatures, especially in deep areas (Forest et al. 2010). Even if primary production increases, the fate of export fluxes will be different on Arctic shelves and over the deep Arctic Ocean (Lalande et al. 2014). It is likely that particulate organic carbon export will remain low above the central basins unless additional nutrients are supplied to surface waters (Codispoti et al. 2013; Lalande et al. 2014). Decreased export of diatoms, and dominance of coccolithophores were observed in Fram Strait during the warm period of 2005-07 (Bauerfeind et al. 2009; Lalande et al. 2013). Warmer water temperature also resulted in lower export fluxes of smaller zooplankton faecal pellets due to a shift in zooplankton community composition towards small-sized zooplankton species or a shift in phytoplankton composition that affected grazing and faecal pellet production (Lalande et al. 2013). However, over the shelves, increased primary productivity will likely supply more food to pelagic and benthic consumers, while the low-productivity zone of the multi-year sea ice would shrink (Bluhm & Gradinger 2008). Sea-ice retreat off the shelves may result in upwelling of nutrients or planktonic prey from the basins onto the shelves (Carmack et al. 2004). Based on the inorganic carbon availability, Anderson & Kaltin (2001) proposed a possible increase of up to 50 g C m-2 integrated over the upper 100 m of the water column across the Eurasian Basin, mainly as a result of ice loss. Nevertheless, it is the availability of nutrients, mainly nitrogen and phosphorous, that ultimately determines the total amount of primary productivity possible in any given ocean (Codispoti et al. 2013).
With reduced ice cover, ice-edge algal blooms will be displaced progressively northwards. Although benthic communities will still receive high-quality food in the short term, if the sea-ice edge retreats past the shelf break, shelf communities will no longer benefit from this early season food source (Renaud, Carroll et al. 2008) which would result in increased food input to slope and deep-sea communities (Carmack & Chapman 2003). Since it is likely that at least a part of deep-sea fauna originates from shelf species and Arctic shelf and deep- sea taxa largely overlap (Bluhm, Ambrose et al. 2011), some shelf species would be able to dwell in the deep sea. If shelf species were unable to leave the slopes or survive in slope or deep-sea habitats, many Arctic shelf-benthos taxa could become locally extinct (Renaud, Carroll et al. 2008). Changes in under-ice community structure were noticed in the Eurasian Basin in geographically close sampling locations collected within short time intervals. Habitat partitioning between sympagic and pelagic species can be abrupt, creating small-scale patterns in the surface layer community according to sea-ice habitat conditions. The difference in ice coverage was accurately mirrored by a conspicuous dominance of the ice- associated amphipod Apherusa glacialis in ice-covered waters, versus a dominance of the pelagic amphipod Themisto libellula in the surface community of ice-free waters (Koszteyn et al. 1995; Hop & Pavlova 2008; David et al. 2015).
Fronts are regions characterized by narrow bands of horizontal gradients in temperature, salinity, density and biological properties that separate broader areas of different vertical structure (Mann & Lazier 1996; Belkin et al. 2003). In the Barents Sea, the PF separates warmer, more saline Atlantic waters in the south from colder, less saline Arctic water in the north (Loeng 1991). Fronts can
play a role in setting surface layer properties by restratifying the surface layer (Timmermans & Winsor 2013) and thereby enhancing primary production. Fronts are known to support elevated biomasses of phytoplankton (Iverson et al. 1979), planktonic organisms (Basedow et al.) and hyper-benthic communities (Dewicke et al. 2002) as well as bird and mammal aggregations (Bluhm et al. 2007). However, a recent study of the PF in the Barents Sea found no stimulatory effect on this front on primary production, and this result is attributed to this front being weak in terms of density (Erga et al. 2014). A related study found high secondary production at the PF, but also in surrounding waters (Basedow et al. 2014). Along with sea temperature rise and increased advection of Atlantic or Pacific waters into the Arctic Ocean, characteristics and location of PF are likely to change, influencing energy transfer to higher trophic levels. Northward displacement of the PF in the Barents Sea was predicted from coupled biophysical model for a B2 Intergovernmental Panel on Climate Change scenario (Huse & Ellingsen 2008). Simulations showed that PF displacement in the Barents Sea had an impact on the distribution and spawning of capelin (Mallotus villosus). The model predicted increased production and large interannual variability in the Barents Sea, characteristic for the MIZ (Wassmann et al. 2010).
Permanent ice cover—high Arctic
We have only limited knowledge of the energy flow and trophic structure of Arctic deep-sea regions. Very little is known about the linkages of the seasonal production pulse to the deep-sea communities in the High Arctic or even about the deep-sea communities themselves. Therefore, many of the assumptions made for shelf Arctic systems may not be valid for the central Arctic. The few available studies from the central Arctic report extremely low species richness and biomass (Kroncke 1994, 1998) for meiofaunal (Vanreusel et al. 2000) and macrofaunal taxa (Kroncke 1994, 1998; Clough et al. 1997; Deubel 2000; Bluhm et al. 2005; Bluhm, Ambrose et al. 2011), as well as a decrease in diversity with increasing water depth (Kroncke et al. 1998). Primary productivity in the central Arctic is limited by light and nutrients. Constrained by light, sea-ice algal primary production occurs only from May to August and may be further restricted by ice thickness and snow cover (Rysgaard et al. 2001; Nicolaus et al. 2012). Nutrient supply on the other hand is constrained by stratification (Bourgain & Gascard 2011) and may not be sufficient in the central Arctic (Tremblay et al. 2008). Estimated average primary production in the ice-covered central Arctic is low, on the order of 1 to 25 g C m-2 y-1 (Wheeler et al. 1997; Wassmann et al. 2010), with ice algae production contributing from 0 to 80% (Gosselin et al. 1997; Wassmann et al. 2008). Bauerfeind et al. (2009) suggested very efficient processing of carbon within the water column, with <10% of primary production reaching the seafloor in the deep sea (Fram Strait). Low primary production and export flux result in low abundance of suspension feeders in the deep basins (Kroncke et al. 1998) and a dominance of deposit feeders (van Oevelen et al. 2011). However, suspension feeders in the deep sea are known to utilize re-suspended material and directly compete with deposit feeders on the scarce food available (Lampitt et al. 1993; Iken et al. 2001). Although most benthic species in the deep sea seem to be able to cope with refractory material (van Oevelen et al. 2011), fresh phytodetritus may also arrive to the seafloor and support suspension feeders and surface deposit feeders such as cumaceans (Iken et al. 2005). Also, Boetius et al. (2013) reported fast response to Melosira falls of opportunistic deep-sea megafauna species, such as the holothurians Kolga hyalina and Elpidia heckeri, and the ophiurid Ophiostriatus striatus. Increased sediment respiration rates showed that sediment bacteria also profited from this ice algae deposition (Boetius et al. 2013). However, infauna were probably unable to utilize ice algae as infaunal burrows and tubes were rarely seen in the under-water video footage, although they are common in other deep-sea basins with seasonally sedimenting phytoplankton blooms (Boetius et al. 2013). In the Arctic deep-sea plains, benthic communities are constrained by strong seasonality and limited food supply (Iken et al. 2005); ice algae production related to permanent sea-ice cover and export fluxes of organic matter to the seafloor are therefore important in these ecosystems. Benthic trophic pathways in the deep Arctic Ocean are longer than on the shelf region or in the temperate deep sea owing to the continuous recycling and thus isotopic enrichment of food particles in the benthic system, while a more direct link to fresh phytodetritus exists in the pelagic system (Iken et al. 2005; Bergmann et al. 2009).
In the past, the central Arctic Ocean has been covered with multi-year ice, but the marked decline in multi-year ice (Maslanik et al. 2011) suggests that the region could be ice-free by the summer of 2040 (Polyakov et al. 2010). Moreover, according to the latest publications, the central Arctic Ocean is no longer covered with multi-year ice (Polyakov et al. 2012) while the remnant multi-year sea ice occurs along the north-west flank of the Canadian Arctic Archipelago, where it can drift southwards, out over the Southern Beaufort Sea and northwards (Barber et al. 2009). If we assume that areas formerly covered
with thick multi-year ice will have a thinner ice cover, permitting higher primary production, we can project a higher annual primary production level due to light availability, provided nutrients are available, which may not be the case in the basins (Codispoti et al. 2013; Matrai et al. 2013). A massive under-ice bloom was reported recently from consolidated ice pack in the northern Chukchi Sea, with phytoplankton biomass beneath the ice being about fourfold greater than in open water (Arrigo et al. 2012). Similar massive blooms might be widespread in the Arctic Ocean, in relation to the lower nutrient levels available. More work is needed to determine the extent to which such blooms are controlled by thinning sea ice and proliferating melt pond fractions and how they affect marine ecosystems.
Although higher light penetration will promote ice algal growth, low level of nutrients available and progressing climate warming may also reduce the algal growing season through increased thermal or haline stratification, limiting mixing and upward nutrient transport resulting in smaller export flux to the seafloor (Carmack & Wassmann 2006; Slagstad et al. 2011). In addition, if zooplankton abundance increases as warmer Atlantic and Pacific waters are transported into the Arctic Ocean (Hirche & Kosobokova 2007), the grazing pressure will increase, leading to increased retention of organic carbon in the water column. Some studies suggest that the flux of ice algae and ice-related particulate organic matter will decrease along with sea-ice retreat and loss of multi-year ice (Forest et al. 2010). This may lead to decreased carbon deposition at the deep seafloor to already food-limited fauna, but these shifts are not expected to be rapid (van Oevelen et al. 2011). Yet, the lack of reliable baseline information makes predictions difficult and identifying change nearly impossible (Wassmann et al. 2011).
Gaps and recommendations
Despite numerous recent studies (Table 2, Fig. 1), major gaps remain in the knowledge of general processes governing biodiversity, food-web structure, trophic transfer efficiency and functioning of Arctic ecosystems. Since different regions of the Arctic have received varying levels of scientific attention (Table 2, Fig. 1), these recommendations may not apply to the whole Arctic and should be treated as more general statements. Given that these processes are not yet clearly quantified, any attempt to project changes that may occur in Arctic food webs in the future, such as that suggested in this review, should be taken cautiously. Most predictions are qualitative and biased towards conditions on the shelves, while quantitative ones remain scarce (but see Zhang et al. 2010; Popova et al. 2012; Slagstad et al. 2011). Since a lot of changes in the Arctic food webs are expected to be driven by a shift from decreasing sea-ice algae and an increase in pelagic production, observational, experimental and modelling approaches of the present-day coupling of these two production pathways in food webs should first be implemented, and subsequently combining different scientific methods will allow for the establishment of projection methodologies for Arctic ecosystems.
Large-scale studies of food webs across Arctic regions highlighting inherent differences among regions are still lacking. Although deep-sea areas are still under-sampled because of the difficulty in accessing field sites, and a consistent sampling design does not exist Arctic-wide, the lack of sampling is not the main issue. Throughout the years data on the response of different benthic communities to climate change and on food webs have increased from multiple projects. These include HAUSGARTEN, the Alfred Wegener Institute's long-term monitoring programme in Fram Strait (Soltwedel et al. 2005), with data over 10 years, and the Distributed Biological Observatory (DBO), an international initiative in the Pacific Arctic (Grebmeier et al. 2010), with data over 30 years, including time series from Long-term Census of the Arctic (RUSALCA) programme. While efforts to sample the Arctic Ocean must and will continue, a primary ambition should be to gather, combine and analyse existing information. Such efforts have been undertaken recently regarding Arctic biodiversity (e.g., Bluhm, Ambrose et al. 2011; Gill et al. 2011; Piepenburg et al. 2011), food sources and trophic interactions, e.g., the Pacific Marine Arctic Regional Synthesis project (PacMARS), although consistent data on food webs (sensu species interactions) are still missing. Similar-format databases should be established, centralized and made easily accessible to scientists at the international level, in order to explore the issues mentioned in this review, such as the PacMARS effort. Then, to promote further consistency of data collection, a standardized data sampling protocol should be implemented when possible to facilitate data gathering and data set use. Designated sampling stations should be sampled throughout months and years to monitor seasonal and long-term changes in biodiversity (Gill et al. 2011). Apart from species identity, functional lifestyle and life-cycle traits should be recorded, which would allow detecting and monitoring changes in ecosystem functioning (Cadotte et al. 2011).
To complement field sampling and database design, empirical manipulations to be performed in situ, in several places in the Arctic, should be encouraged. Such studies should address quantitative aspects of the impact of global warming on food-web length and components, including primary production rates, grazing rates and growth rates at higher trophic levels, in order to estimate changes in trophic transfer efficiency in Arctic food webs. Studies of diets relative to availability of different foods are also critical to food-web analyses, especially to predicting response to changing conditions. In situ and laboratory experiments could be complemented by use of numerical modelling. Trophic network modelling methods that are widely used in other ecosystems could be implemented in the Arctic to understand food-web structure, the effects of external threats (e.g., increased fisheries pressure, invasive species) on food-web dynamics, and to quantify energy transfers between trophic levels, in conjunction with empirical work (e.g., van Oevelen et al. 2011). Results can be subsequently used to fuel models (e.g., provide nutrient-phytoplankton-zooplankton model parameters). Since data are still lacking, this could be achieved by using well-established predesigned software able to deal with missing parameters such as Ecopath (Pauly et al. 2000), especially for fishery scenarios, and the linear inverse model package LIM in the R statistical software package (van Oevelen et al. 2010). These models have the advantage of including both pelagic and benthic compartments and are able to quantify the strength of pelagic-benthic coupling. Although such models have been implemented for some Arctic regions (e.g., Trites et al. 1999; Dommasnes et al. 2001; Pedersen & Zeller 2001; Aydin et al. 2002; Whitehouse et al. 2014), they need to be updated, integrated and compared, especially after the recent environmental changes that have occurred in the Arctic. Stability analysis methods can also be coupled with the mass-balance modelling framework to provide insights into Arctic food-web structure and intrinsic properties (Neutel et al. 2002). Such methods may comprise analyses of stable-states of population or community dynamics, and analyses of food-web properties through determination of eigenvectors, resilience and persistence levels, and equilibrium shifts. At the theoretical level, population, metapopulation, community and metacommunity dynamic models can be implemented to understand how intra- and interspecific interactions and connectivity affect diversity at different spatial scales, such as between the different Arctic regions and the surrounding oceans (Carr et al. 2011; Hardy et al. 2011). Predicted ice melt and changing hydrodynamics may alter connectivity between distant Arctic populations, in turn affecting community composition and food-web structure. Such advances could provide valuable contributions to predicting future trends of biodiversity and food-web structure in the Arctic.
Statistical modelling, particularly quantification of species-environment relationships at large scales, should be assessed once data are pooled across Arctic regions. The statistical methods developed to date and applied to different biological organisms are an efficient means to disentangle the effects of environmental gradients (both in space and time) on community structure (Dray et al. 2012). Even though these methods are often based on species, they can be applied to functional groups to assess trait-environment relationships, reinforcing the need to monitor species traits during field sampling. Since environmental changes in the Arctic may affect species traits, the whole ecosystem functioning may be altered, and studying functional traits is a good way to assess ecosystem functioning and trophic transfer efficiency (McGill et al. 2006; Cadotte et al. 2011).
Conclusions: winners and losers
Arctic marine ecosystems are currently subjected to accelerating climate warming and fast progressing sea-ice retreat. Although our knowledge of ecosystem functioning and processes still has significant gaps, and the scale and magnitude of climate change remain largely unknown, some qualitative predictions on the fate of Arctic marine food webs are possible. In very general terms, among the "winners" will likely be boreal species as their populations tend to follow increasing sea temperature by shifting their ranges northward (and likely losing ground in the south), and pelagic species, mainly zooplankton, whose abundance and biomass may increase with increasing water column primary production related to more and earlier open water over the Arctic shelves. Pelagic feeding animals, like some fish, marine mammals and seabirds may consequently increase in abundance. Species classified as generalists are more likely to adapt to new conditions than specialists.
Groups that will likely be among the "losers" include Arctic species, especially those that are ice-dependent, as they will be most strongly affected by rising temperatures and diminishing habitat. Benthic species may decrease in biomass with increased pelagic grazing and recycling in the water column, which may lead to reduced amount or quality of organic matter settling from the water column to the seafloor. This will affect benthic feeding marine mammals and seabirds, whose foraging areas will become less productive and prey less available. Food webs will likely lengthen at the low trophic levels, lowering trophic transfer efficiency and thereby lowering the percentage of primary production that reaches top predators. Since many species at the base of the Arctic food webs are seaice dependent, the stability of food webs will likely be negatively affected in areas where trophic redundancy is low. Collaborative multidisciplinary research is necessary
if we are to fully understand the processes and linkages between Arctic marine environments and their associated food webs in the face of a changing North.
Acknowledgements
This publication is an output of the first joint Arctic in Rapid Transition (ART) and Association of Polar Early Career Scientists science workshop, in Sopot, Poland, 23-26 October 2012. This cross-cutting initiative was generously supported by the International Arctic Science Committee, the Prince Albert П of Monaco Foundation, the Polish Academy of Sciences and Institute of Oceanology, the Polish Scientific Committee on Oceanic Research, the International Arctic Research Center, the French National Center for Scientific Research-Laval Takuvik Joint Laboratory, the Alfred Wegener Institute for Polar and Marine Research and the Department of Fisheries and Oceans Canada. The support and initiative of ART is gratefully acknowledged. Financial support for preparation of individual components of this review was provided by the following sources: National Science Centre grant no. DEC-2013/08/M/NZ8/00592 for MK; Resources Aquatiques Quebec and the Natural Sciences and Engineering Research Council of Canada for CM; a Natural Sciences and Engineering Research Council of Canada (NSERC) postgraduate scholarship and W. Garfield Weston Award Foundation for ESC; grants from the US National Science Foundation (NSF-DPP-1204082) and the National Oceanic and Atmospheric Administration (NA08OAR4310608) for JMG; the Partnership for Observations of the Global Oceans-SCOR fellowship programme 2013 and President's grant MK-128.2014.1 and Russian Foundation for Basic Research grant no. 14-01-31053 for DK. SD would like to acknowledge Dr Greg Thiemann, NSERC, Environment Canada and the Molson Foundation.
References
- Amante C. & Eakins B.W. 2009. ET0P01 1 arc-minute global relief model: procedures, data sources and analysis. Pp. 1-19. NOAA Technical Memorandum NESDIS NGDC-24. Boulder, CO: National Oceanic and Atmospheric Administration.
- Ambrose W.G. Jr. & Renaud P.E. 1995. Benthic response to water column productivity patterns: evidence for benthic- pelagic coupling in the Northeast Water Polynya. Journal of Geophysical Research—Oceans 100, 4411-4421.
- Ambrose W.G. Jr. & Renaud P.E. 1997. Does a pulsed food supply to the benthos affect polychaete recruitment patterns in the Northeast Water Polynya? Journal of Marine Systems 10, 483-495.
- Anderson L.G. & Kaltin S. 2001. Carbon fluxes in the Arctic Ocean—potential impact by climate change. Polar Research 20, 225-232.
- Arrigo K.R., Perovich D.K., Pickart R.S., Brown Z.W., van Dijken G.L., Lowry K.E., Mills M.M., Palmer M.A., Balch W.M., Bahr F., Bates N.R., Benitez-Nelson C., Bowler 2012 , Brownlee E., Ehn J.K., Frey K.E., Garley R., Laney S.R., Lubelczyk L., Mathis J., Matsuoka A., Mitchell B.G., Moore G.W.K., Ortega-Retuerta E., Pal S., Polashenski C.M., Reynolds R.A., Schieber B., Sosik H.M., Stephens M. & Swift J.H. 2012. Massive phytoplankton blooms under Arctic sea ice. Science 336,
- Arrigo K.R., Perovich D.K., Pickart R.S., Brown Z.W., van Dijken G.L., Lowry K.A., Mills M.M., Palmer M.A., Balch W.M., Bates N.R., Benitez-Nelson C.R., Brownlee E., Frey K.E., Laney S.R., Mathis J., Matsuoka A., Mitchell 2014 , Moore G.W.K., Reynolds R.A., Sosik H.M. & Swift J.H. 2014. Phytoplankton blooms beneath the sea ice in the Chukchi Sea. Deep-Sea Research Part 11105, 85-104.
- Arrigo K.R. & van Dijken G.L. 2011. Secular trends in Arctic Ocean net primary production. Journal of Geophysical Research—Oceans 116, C09011, doi: 10.1029/2011JC007151.
- Arrigo K.R., van Dijken G. & Pabi S. 2008. Impact of a shrinking Arctic ice cover on marine primary production. Geophysical Research Letters 35, L19603, doi: 10.1029/ 2008GL03 5028.
- Auel H., Haijes M., da Rocha R., Stubing D. & Hagen W. 2002. Lipid biomarkers indicate different ecological niches and trophic relationships of the Arctic hyperiid amphipods, Themisto abyssorum and T. libellula. Polar Biology 25, 374-383.
- Aydin K. & Mueter F. 2007. The Bering Sea—a dynamic food web perspective. Deep-Sea Research Part II54, 2501-2525.
- Aydin K.Y., Lapko V.V., Radchenko V.I. & Livingston P.A. 2002. A comparison of the eastern Bering and western Bering Sea shelf and slope ecosystems through the use of mass-balance food web models. NOAA Technical Memorandum NMFS-APSC-130. Seattle, WA: National Oceanoic and Atmospheric Administration.
- В achy C., L6pez-Garda P., Vereshchaka A. & Moreira D. 2011. Diversity and vertical distribution of microbial eukaryotes in the snow, sea ice and seawater near the north pole at the end of the polar night. Frontiers in Microbiology 2, article no. 106, doi: 10.3389/fmicb.2011.00106.
- Barber D.G., Galley R., Asplin M.G., De Abreu R., Warner K.A., Гибко M., Gupta M., Prinsenberg S. & Julien S. 2009. Perennial pack ice in the southern Beaufort Sea was not as it appeared in the summer of 2009. Geophysical Research Letters 36, L24501, doi: 10.1029/2009GL041434.
- Basedow S.L., Zhou M. & Tande K.S. 2014. Secondary production at the Polar Front, Barents Sea, August 2007. Journal of Marine Systems 130, 147-159.
- Bauerfeind E., Nothig E.M., Beszczyriska A., Fahl K., Kaleschke L., Kreker K., Klages M., Soltwedel T., Lorenzen 2009 & Wegner J. 2009. Particle sedimentation patterns in the eastern Fram Strait during 2000-2005: results from the Arctic long-term observatory HAUS GARTEN. Deep-Sea Research Part 156, 1471-1487.
- Belkin I.M., Comilion P. & Ullman D. 2003. Ocean fronts around Alaska from satellite SST data. In: Proceedings of the American Meteorological Society’s 7th Conference on the Polar Meteorology and Oceanography and Joint Symposium on High- Latitude Climate Variations, 12-16 May, Hyannis, MA. Paper 12.7. Boston: American Meteorological Society.
- Berge J., Heggland K., Bonne O.J., Cottier F., Hop H., Gabrielsen G.W, Nottestad L. & Misund O.A. 2015. First records of Atlantic mackerel (Scomber scombrus) from the Svalbard Archipelago, Norway, with possible explanations for the extension of its distribution. Arctic 68, 54-61.
- Berge J., Johnsen G., Nilsen F., Gulliksen B. & Slagstad D. 2005. Ocean temperature oscillations enable reappearance of blue mussels Mytilus edulis in Svalbard after 1000 years of absence. Marine Ecology Progress Series 303, 167-175.
- Bergmann M., Dannheim J., Bauerfeind E. & Klages M. 2009. Trophic relationships along a bathymetric gradient at the deep-sea observatory HAUSGARTEN. Deep-Sea Research Part 156, 408-424.
- Birdlife International. 2012a. Alle alle. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet at www.iucnredlist.org on 3 July 2014.
- Birdlife International. 2012b. Pulmarus glacialis. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet at www.iucnredlist.org on 3 July 2014.
- Birdlife International. 2012c. Somateriafischeri. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet at www.iucnredlist.org on 3 July 2014.
- Blake J.A. 1993. Life history analysis of five dominant infaunal polychaete species from the continental slope off North Carolina. Journal of Marine Biology Association of United Kingdom 73, 123-141.
- Blanchard A.L., Feder H.M. & Hoberg M.K. 2010. Temporal variability of benthic communities in an Alaskan glacial fjord, 1971-2007. Marine Environmental Research 69, 95-107.
- Bluhm B., MacDonald I.R., Debenham C. & Iken K. 2005. Macro- and megabenthic communities in the High Arctic Canada Basin: initial findings. Polar Biology 28, 218-231.
- Bluhm B.A., Ambrose W.G. Jr., Bergmann M., Clough L.M., Gebruk A.V, Hasemann C., Iken K., Klages M., MacDonald I.R., Renaud P.E., Schewe I., Soltwedel T. & Wlodarska- Kowalczuk M. 2011. Diversity of the Arctic deep-sea benthos. Marine Biodiversity 41, 87-107.
- Bluhm B.A., Coyle K.O., Konar B. & Highsmith R.C. 2007. High gray whale relative abundances associated with an oceanographic front in the south-central Chukchi Sea. Deep-Sea Research Part II54, 2919-2933.
- Bluhm B.A. & Gradinger R. 2008. Regional variability in food availability for Arctic marine mammals. Ecological Applications 18, 77-96.
- Bluhm B.A., Gradinger R. & Hopcroft R.R. 2011. Arctic ocean diversity synthesis. Marine Biodiveristy 41, 1-4.
- Boetius A., Albrecht S., Bakker K., Bienhold C., Felden J., Fernandez-Mendez M., Hendricks S., Katlein C., Lalande C., Krumpen T, Nicolaus M., Peeken I., Rabe B., Rogacheva A., Rybakova E., Somavilla R. & Wenzhofer F. 2013. Export of algal biomass from the melting Arctic sea ice. Science 339, 1430-1432.
- Bourgain P. & Gascard J.C. 2011. The Arctic Ocean halocline and its interannual variability from 1997 to 2008. Deep-Sea Research Part 158, 745-756.
- Cadotte M., Carscadden K. & Mirotchnick N. 2011. Beyond species: functional diversity and the maintenance of ecological processes and services. Journal of Applied Ecology 48, 1079-1087.
- Campbell R.G., Sherr E.B., Ashjian C.J., Plourde S., Sherr 2009, Hill V. & Stochwell D.A. 2009. Mesozooplankton prey preference and grazing impact in the western Arctic Ocean. Deep-Sea Research Part II56, 1274-1289.
- Carmack E. & Chapman D.C. 2003. Wind-driven shelf/basin exchange on an Arctic shelf: the joint roles of ice cover extent and shelf-break bathymetry. Geophysical Research Letters 30, 1778.
- Carmack E. & Wassmann P. 2006. Food webs and physical- biological coupling on pan-Arctic shelves: unifying concepts and comprehensive perspectives. Progress in Oceanography 71, 446-477.
- Carmack E.C., Macdonald R.W. & Jasper S. 2004. Phytoplankton productivity on the Canadian shelf of the Beaufort Sea. Marine Ecology Progress Series 277, 37-50.
- Carr C.M., Hardy S.M., Brown T.M., Macdonald TA. & Hebert P.D.N. 2011. A tri-oceanic perspective: DNA barcoding reveals geographic structure and cryptic diversity in Canadian polychaetes. PLoS One 6, e22232, doi: 10.1371/joumal. pone.0022232.
- Chambouvet A., Morin P, Marie D. & Guillou L. 2008. Control of toxic marine dinoflagellates blooms by serial parasitic killers. Science 322, 1254-1257.
- Clarke A. & Harris C.M. 2003. Polar marine ecosystems: major threats and future change. Environmental Conservation 30, 1-25.
- Clough L.M., Ambrose W.G. Jr., Cochran J.K., Barnes C., Renaud PE. & Aller R.C. 1997. Infaunal density, biomass and bioturbation in the sediments of the Arctic Ocean. Deep-Sea Research Part II44, 1683-1704.
- Clough L.M., Renaud P.E. & Ambrose W.G. Jr. 2005. Impacts of water depth, sediment pigment concentration, and benthic macrofaunal biomass on sediment oxygen demand in the western Arctic Ocean. Canadian Journal of Fisheries and Aquatic Sciences 62, 1756-1765.
- Codispoti L.A., Kelly V, Thessen A., Matrai P., Suttles S., Hill V, Steele M. & Light B. 2013. Synthesis of primary production in the Arctic Ocean: HI. Nitrate and phosphate based estimates of net community production. Progress in Oceanography 110, 126-150.
- Collins R.E. & Deming J.W. 2011. Abundant dissolved genetic material in Arctic sea ice. Part II: viral dynamics during autumn freeze-up. Polar Biology 34, 1831-1841.
- Comeau A.M., Li W.K., Tremblay J.E., Carmack E. & Lovejoy 2011 Arctic Ocean microbial community structure before and after the 2007 record sea ice minimum. PLoS One 6, e27492. doi: 10.1371/journal.pone.0027492.
- Comeau A.M., Philippe B., Thaler M., Gosselin M., Poulin M. & Lovejoy C. 2013. Protists in Arctic drift and land-fast ice. Journal of Phycology 49, 229-240.
- Comiso J.C. 2012. Large decadal decline of the Arctic multiyear ice cover. Journal of Climate 25, 1176-1193.
- Cooper L.W, Grebmeier J.M., Larsen I.L., Egorov V.G., Theodorakis C., Kelly H.P. & Lovvom J. 2002. Seasonal variation in sedimentation of organic materials in the St. Lawrence Island polynya region, Bering Sea. Marine Ecology Progress Series 226, 13-26.
- Daase M., Verpe 0. & Falk-Petersen S. 2014. Non-consumptive mortality in copepods: occurrence of Calanus spp. carcasses in the Arctic Ocean during winter. Journal of Plankton Research 36, 129-144.
- Damis G., Robert D., Pomerlau C., Link H., Archambault P., Nelson R.J., Geoffroy M., Tremblay J.E., Lovejoy C., Ferguson S., Hunt B.V. & Fortier L. 2012. Current state and trends in Canadian Arctic marine ecosystems: П. Heterotrophic food web, pelagic-benthic coupling, and biodiversity. Climatic Change 115, 179-205.
- Daufresne M., Lengfellener K. & Sommer U. 2009. Global warming benefits the small in aquatic ecosystems. Proceedings of the National Academy of Sciences of the United States of America 106, 12788-12793.
- David C., Lange B., Rabe B. & Flores H. 2015. Community structure of under-ice fauna in the Eurasian central Arctic Ocean in relation to environmental properties of sea-ice habitats. Marine Ecology Progress Series 522, 15-32.
- De Laender F., Van Oevelen D., Soetaert K. & Middelburg J.J. 2010. Carbon transfer in herbivore- and microbial loop- dominated pelagic food webs in the southern Barents Sea during spring and summer. Marine Ecology Progress Series 398, 93-107.
- Deubel H. 2000. Struktureigenschaften und Nahrungsbedarf der Zoobenthosgemeinschaften im Bereich des Lomonossowrilckens im Arktischen Ozean. (Structures and nutrition requirements of macrozoobenthic communities in the area of the Lomonossov Ridge in the Arctic Ocean.) Berichte zur Polar- und Meeresforschung 370. Bremerhaven: Alfred Wegener Institute.
- Dewicke A., Rottiers V, Mees J. & Vincx M. 2002. Evidence for an enriched hyperbenthic fauna in the Frisian Front (North Sea). Journal of Sea Research 47, 121-139.
- Dommasnes A., Christensen V, Eilertsen B., Kvamme C., Melle W, Nottestad L., Pedersen T., Tjelmeland S. & Zeller 2001 An ecopath model for the Norwegian Sea and Barents Sea. In S. Guenette et al. (eds.): Fisheries impacts on North Atlantic ecosystems: models and analyses. Fisheries Centre Research Reports. Pp. 213-240. Vancouver: Fisheries Centre, University of British Columbia.
- Dray S., Pelissier R., Couteron P., Fortin M.J., Legendre P., Peres-Neto PR., Bellier E., Bivand R., Blanchet F.G., De Caceres M., Dufour A.B., Heegaard E., Jombart T., Munoz 2012 , Oksanen J., Thiouloise J. & Wagner H.H. 2012. Community ecology in the age of multivariate multiscale spatial analysis. Ecological Monographs 82, 257-275.
- Duarte C.M. 2007. Marine ecology warms up to theory. Trends in Ecology and Evolution 22, 331-333.
- Dunbar R.B. & Leventer A.R. 1987. Sediment fluxes beneath fast ice: October 19 86 through February 1987. Antarctic Journal of the United States 22, 112-115.
- Dunton K.H., Schonberg S.V & Cooper L.W 2012. Food web structure of the Alaskan nearshore shelf and estuarine lagoons of the Beaufort Sea. Estuaries and Coasts 35, 416-435.
- Dunton K.H., Weingartner T. & Carmack E.C. 2006. The nearshore western Beaufort Sea ecosystem: circulation and importance of terrestrial carbon in Arctic coastal food webs. Progress in Oceanography 71, 362-378.
- Erga S.R., Ssebiyonga N., Hamre B., Frette 0., Rey F. & Drinkwater K. 2014. Nutrients and phytoplankton biomass distribution and activity at the Barents Sea Polar Front during summer near Hopen and Storbanken. Journal of Marine Systems 130, 181-192.
- Falkowski P.G., Barber R.T. & Smetacek V. 1998. Biogeochemical controls and feedbacks on ocean primary production. Science 281, 200-206.
- Falk-Petersen S., Mayzaud P., Kattner G. & Sargent J. 2009. Lipids and life strategy of Arctic Calanus. Marine Biology Research 5, 18-39.
- Falk-Petersen S., Pavlov V, Timofeev S. & Sargent J.R. 2006. Climate variability and possible effects on Arctic food chains: the role of Calanus. In J.B. 0rbaek et al. (eds.): Arctic alpine ecosystems and people in a changing environment. Pp. 147-166. Berlin: Springer.
- Falk-Petersen S., Sargent J.R., Lonne O.J. & Timofeev S. 1999. Functional biodiversity in lipids of Antarctic zooplankton: Calanoides acutus, Calanus propinquus, Thysanoessa macrura and Euphausia crystallorophias. Polar Biology 21, 37-47.
- Feder H.M., Iken K., Blanchard A.L., Jewett S.C. & Schonberg S. 2011. Benthic food web structure in the southemeastem Chukchi Sea: an assessment using 513C and S15N analyses. Polar Biology 34, 521-532.
- Feder H.M., Jewett S.C. & Blanchard A. 2007. Southeastern Chukchi Sea (Alaska) macrobenthos. Polar Biology 30, 261-275.
- Fetterer F., Knowles K., Meier W. & Savoie M. 2002 (updated. Sea ice index. G02135. Boulder, CO: National Snow and Ice Data Center. Accessed on the internet at http://dx.doi.org/10.7265/N5QJ7F7W on 07.07.2014
- Forest A., Wassmann P., Slagstad D., Bauerfeind E., Noting E.M. & Klages M. 2010. Relationships between primary production and vertical particle export at the Atlantic-Arctic boundary (Fram Strait, HAUSGARTEN). Polar Biology 33, 1733-1746.
- Fortier M., Fortier L., Michel C. & Legendre L. 2002. Climatic and biological forcing of the vertical flux of biogenic particles under seasonal Arctic sea ice. Marine Ecology Progress Series 225, 1-16.
- Fortier L., Pascal S., Josee M. & Barber D. 2006. Survival of Arctic cod larvae (Boreogadus saida) in relation to sea ice and temperature in the Northeast Water Polynya (Greenland Sea). Canadian Journal of Fisheries and Aquatic Sciences 63, 1608-1616.
- Frey K.E. & McClelland J.W. 2009. Impacts of permafrost degradation on Arctic river biogeochemistry. Hydrological Processes 23, 169-182.
- Frey K.E., McClelland J.W., Holmes R.M. & Smith L.C. 2007. Impacts of climate warming and permafrost thaw on the riverine transport of nitrogen and phosphorus to the Kara Sea. Journal of Geophysical Research—Biogeosciences 112, G04S58, doi: 1O.1O29/2OO6JGOOO369.
- Gill M.J., Crane K., Hindrum R., Ameberg P., Bysveen I., Denisenko N.V., GofmanV., Grant-Friedman A., Gudmundsson 2011, Hopcroft R.R., Iken K., Labansen A., Liubina O.S., Melnikov LA., Moore S.E., Reist J.D., Sirenko B.I., Stow J., Ugarte F., Vongraven D. & Watkins J. 2011. Arctic Marine Biodiversity Monitoring Plan (CBMP-MARINE PLAN). CAFF Monitoring Series Report 3. Akureyri: Conservation of Arctic Flora and Fauna International Secretariat.
- Gosselin M., Lecasseur M., Wheeler P.A., Homer R.A. & Booth B.C. 1997. New measurements of phytoplankton and ice algal production in the Arctic Ocean. Deep-Sea Research Part 1144, 1623-1644.
- Gradinger R. 1995. Climate change and biological oceanography of the Arctic Ocean. Philosophical Transactions of the Royal Society of London Series A 352, 277-286.
- Gradinger R. 2009. Sea ice algae: major contributors to primary production and algal biomass in the Chukchi and Beaufort seas during May/June 2002. Deep-Sea Research Part 1156, 1201-1212.
- Gradinger R. & Bluhm B. 2004. In situ observations on the distribution and behavior of amphipods and Arctic cod (Boreogadus saida) under the sea ice of the High Arctic Canadian Basin. Polar Biology 27, 595-603.
- Grebmeier J.M. 2012. Shifting patterns of life in the Pacific Arctic and sub-Arctic seas. Annual Review of Marine Science 4, 63-78.
- Grebmeier J.M. & Barry J.P. 2007. Benthic processes in polynyas. In W. Smith Jr. & D. Barber (eds.): Polynyas: windows to the world. Pp. 363-390. Amsterdam: Elsevier.
- Grebmeier J.M., Cooper L.W, Feder H.M. & Sirenko B.I. 2006. Ecosystem dynamics of the Pacific-influenced northern Bering and Chukchi seas in the Amerasian Arctic. Progress in Oceanography 71, 331-361.
- Grebmeier J.M. & McRoy C.P. 1989. Pelagic-benthic coupling on the shelf of the northern Bering and Chukchi seas. III. Benthic food supply and carbon cycling. Marine Ecology Progress Series 53, 79-91.
- Grebmeier J.M., McRoy C.P. & Feder H.M. 1988. Pelagic- benthic coupling on the shelf of the northern Bering and Chukchi seas. I. Food supply source and benthic biomass. Marine Ecology Progress Series 48, 57-67.
- Grebmeier J.M., Moore S.E., Overland J.E., Frey K.E. & Gradinger R. 2010. Biological response to recent pacific Arctic sea ice retreats. Eos, Transactions of the American Geophysical Union 91, 161-162.
- Grebmeier J.M., Overland J., Moore S.E., Farley E.V., Carmack 2006., Cooper L.W, Frey K.E., Helle J.H., McLaughlin F.A. & McNutt S.L. 2006. A major ecosystem shift in the northern Bering Sea. Science 311, 1461-1464.
- Gupta M., Barber D.G., Scharien R.K. & Isleifson D. 2014. Detection and classification of surface roughness in an Arctic marginal sea ice zone. Hydrological Processes 28, 599-609.
- Hansell D.A. & Ducklow H.W. 2003. Bacterioplankton distribution and production in the bathypelagic ocean: directly coupled to particulate organic carbon export? Limnology and Oceanography 48, 150-156.
- Hardy S.M., Carr C.M., Hardman M., Steinke D., Corstorphine E. & Mah C. 2011. Biodiversity and phylogeography of Arctic marine fauna: insights from molecular tool. Marine Biodiversity 41, 195-210.
- Hegseth E.N. 1998. Primary production of the northern Barents Sea. Polar Research 17, 113-123.
- Herbert R.J.H., Southward A.J., Sheader M. & Hawkins S.J. 2007. Influence of recruitment and temperature on distribution of intertidal barnacles in the English Channel. Journal of the Marine Biological Association of the United Kingdom 87, 487-499.
- Higdon J.W. & Ferguson S. 2009. Loss of Arctic sea ice causing punctuated change in sightings of killer whales (Orcinus orca) over the past century. Ecological Applications 19, 1365- 1375.
- Higgins M.E. & Cassano J.J. 2009. Impacts of reduced sea ice on winter Arctic atmospheric circulation, precipitation and temperature. Journal of Geophysical Research—Atmospheres 114, D16107, doi: 10.1029/2009JD011884.
- Hirche H.J. & Kosobokova K.N. 2007. Distribution of Calanus flnmarchicus in the northern North Atlantic and Arctic Ocean—expatriation and potential colonization. Deep-Sea Research Part П 54, 2729-2747.
- Hobbs R.J., Arico S., Aronson J., Baron J.S., Bridgewater P., Cramer V.A., Epstein PR., Ewel J.J., Klink C.A., Lugo A.E., Norton D., Ojima D., Richardson D.M., Sanderson E.W, Valladeres F., Vila M., Zamora R. & Zobel M. 2006. Novel ecosystems: theoretical and management aspects of the new ecological world order. Global Ecology and Biogeography 15, 1-7.
- Hobson K.A., Ambrose WG. Jr. & Renaud P.E. 1995. Sources of primary production, benthic-pelagic coupling, and throphic relationships within the Northeast Water Polynya: insights from 513Cand S15N analysis. Marine Ecology Progress Series 128, 1-10.
- Hodges L.R., В ano N., Hollibaugh J.T. & Yager P.L. 2005. Illustrating the importance of particulate organic matter to pelagic microbial abundance and community structure—an Arctic case study. Aquatic Microbial Ecology 40, 217-227.
- Hollowed A.B., Planque B. & Loeng H. 2013. Potential movement of fish and shellfish stocks from the sub-Arctic to the Arctic Ocean. Fisheries Oceanography 22, 355-370.
- Hop H. & Gjosaeter H. 2013. Polar cod (Boreogadus saida) and capelin (Mallotus villosus) as key species in marine food webs of the Arctic and the Barents Sea. Marine Biology Research 9, 878-894.
- Hop H. & Pavlova O. 2008. Distribution and biomass transport of ice amphipods in drifting sea ice around Svalbard. Deep- Sea Research Part II55, 2292-2307.
- Howard-Jones M.H., Ballard V.D., Allen A.E., Frischer M.E. & Verity P.G. 2002. Distribution of bacterial biomass and activity in the Marginal Ice Zone of the central Barents Sea during summer. Journal of Marine Systems 38, 77-91.
- Hunt G.L. Jr., Blanchard A., Boveng P., Dalpadado P., Drinkwater K., Eisner L., Hopcroft R.R., Kovacs K., Norcross B.L., Renaud P.E., Reigstad M., Renner M., Skjoldal H.R., Whitehause A. & Woodgate R. 2013. The Barents and Chukchi seas: comparison of two Arctic shelf ecosystems. Journal of Marine Science 109-110, 43-68.
- Hunt G.L. Jr & Stabeno P.J. 2002. Climate change and the control of energy flow in the southeastern Bering Sea. Progress in Oceanography 55, 5-22.
- Huse G. & Ellingsen I. 2008. Capelin migrations and climate change—a modelling analysis. Climatic Change 87, 177-197.
- Iken K., Bluhm B. & Dunton K. 2010. Benthic food-web structure under differing water mass properties in the southern Chukchi Sea. Deep-Sea Research Part 1157, 71-85.
- Iken K., Bluhm B.A. & Gradinger R. 2005. Food web structure in the High Arctic Canada basin: evidence from 513C and S15N analysis. Polar Biology 28, 238-249.
- Iken К., Brey T., Wand U., Voigt J. & Junghans P. 2001. Food web structure of the benthic community at the Porcupine Abyssal Plain (NE Atlantic): a stable isotope analysis. Progress in Oceanography 50, 383-405.
- Iverson R.L., Whitledge T.E. & Goering J.J. 1979. Fine- structure of chlorophyll and nitrate in the southeastern Bering Sea shelf break front. Nature 281, 664-666.
- Jefferson T.A., Karkzmarski L., Laidre K., O'Corry-Crowe G., Reeves R., Rojas-Bracho L., Secchi E., Slooten E., Smith B.D., Wang J.Y. & Zhou K. 2012. Delphinapterus leucas. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet at www.iucnredlist.org on 3 July 2014
- Ji R., Ashpan C.J., Campbell R.G., Chen C., Gao G., Davis C.S., Cowles G.W & Beardsley R.C. 2012. Life history and biogeography of Calarms copepods in the Arctic Ocean: an individual-based modeling study. Progress in Oceanography 96, 40-56.
- Ji R., Jin M. & Varpe O. 2013. Sea ice phenology and timing of primary production pulses in the Arctic Ocean. Global Change Biology 19, 734-741.
- Kfdra M., Kulinski K., Walkusz W. & Legezynska J. 2012. The shallow benthic food web structure in the High Arctic does not follow seasonal changes in the surrounding environment. Estuarine, Coastal and Shelf Science 114, 183-191.
- Kfdra M., Renaud P.E., Andrade H., Goszczko I. & Ambrose WG. Jr. 2013. Benthic community structure, diversity, and productivity in the shallow Barents Sea bank (Svalbard Bank). Marine Biology 160, 805-819.
- Kirchman D.L., Hill V, Cottrell M.T., Gradinger R., Malmstron R.R. & Parker A. 2009. Standing stocks, production, and respiration of phytoplankton and heterotrophic bacteria in the western Arctic Ocean. Deep-Sea Research Part II 56, 1237-1248.
- Koszteyn J., Timofeev S., Weslawski J.M. & Malinga B. 1995. Size structure of Themisto abyssorum (Boeck) and Themisto libellula (Mandt) populations in European Arctic seas. Polar Biology 15, 85-92.
- Kovacs K. 2008. Pagophilus groenlandicus. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet at www.iucnredlist.org on 3 July 2014
- Kovacs K.M. 2002. Bearded seal Erignathus barbatus. In WF. Perrin et al. (eds.): Encyclopedia of marine mammals. Pp. 84- 87. San Diego, CA: Academic Press.
- Kovacs K.M. & Lydersen C. 2008. Climate change impacts on seals and whales in the North Atlantic Arctic and adjacent shelf seas. Science Progress 91, 117-150.
- Kovacs K.M., Moore S., Lydersen C. & Overland J.E. 2011. Impacts of changing sea ice conditions on Arctic marine mammals. Marine Biodiversity 41, 181-194.
- Kozlovsky D.G. 1968. A critical evaluation of the trophic level concept. I. Ecological efficiencies. Ecology 49, 48-60.
- Kritzberg E.S., Duarte C.M. & Wassmann P. 2010. Changes in Arctic marine bacterial carbon metabolism in response to increasing temperature. Polar Biology 33, 1673-1682.
- Kroncke I. 1994. Macrobenthos composition, abundance and biomass in the Arctic Ocean along a transect between Svalbard and the Makarov Basin. Polar Biology 14, 519-529.
- Kroncke I. 1998. Macrofauna communities in the Amudsen Basin, at the Morris Jesup Rise and at the Yermak Plateau (Eurasian Arctic Ocean). Polar Biology 19, 383-392.
- KrOncke I., Dippner J.W, Heyen H. & Zeiss B. 1998. Longterm changes in macrofaunal communities off Norderney (East Frisia, Germany) in relation to climate variability. Marine Ecology Progress Series 167, 25-36.
- Kubiszyn A.M., Piwosz K., Wiktor J.M. Jr. & Wiktor J.M. 2014. The effect of inter-annual Atlantic water inflow variability on the planktonic protist community structure in the west Spitsbergen waters during the summer. Journal of Plankton Research 36, 1190-1203.
- Kulinski K., Ktzdra M., Legezynska J., Ghichowska M. & Zaborska A. 2014. Particulate organic matter sinks and sources in High Arctic fjord. Journal of Marine Systems 139, 27-37.
- Laidre K.L., Stirling I., Lowry L.F., Wiig 0., Heide-Jorgensen M.P. & Ferguson S.H. 2008. Quantifying the sensitivity of Arctic marine mammals to climate-induced habitat change. Ecological Applications 18, S97-S125.
- Lalande C., Bauerfeind E., Noting E. & Beszczynska-Moller A. 2013. Impact of a warm anomaly on export fluxes of biogenic matter in the eastern Fram Strait. Progress in Oceanography 109, 70-77.
- Lalande C., Grebmeier J.M., Wassmann P., Cooper L.W, Flint M.V & Sergeeva V.M. 2007. Export fluxes of biogenic matter in the presence and absence of seasonal sea ice cover in the Chukchi Sea. Continental Shelf Research 27, 2051-2065.
- Lalande C., Noting E.M., Soma villa R., Bauerfeind E., Shevchenko V. & Okolodkov Y. 2014. Variability in underice export fluxes of biogenic matter in the Arctic Ocean. Global Biogeochemical Cycles 28, 571-583.
- Lampitt R.S., Hillier WR. & Challenor P.G. 1993. Seasonal and dial variation in the open ocean concentration of marine snow aggregates. Nature 362, 737-739.
- Layman C.A., Arrington D.A., Montafla C.G. & Post D.M. 2007. Can stable isotope ratios provide for community-wide measures of trophic structure? Ecology 88, 42-48.
- Lee S.H., Whitledge T.E. & Kang S.H. 2007. Recent carbon and nitrogen uptake rates of phytoplankton in Bering Strait and the Chukchi Sea. Continental Shelf Research 27, 2231-2249.
- Leu E., Soreide J., Hessen D.O., Falk-Petersen S. & Berge J. 2011. Consequences of changing sea-ice cover for primary and secondary producers in the European Arctic shelf seas: timing, quantity, and quality. Progress in Oceanography 90, 18-32.
- Lewis J.R. 1996. Coastal benthos and global wanning: strategies and problems. Marine Pollution Bulletin 32, 698-700.
- Lewis O.T. 2005. Climate change, spedes-area curves and the extinction crisis. Philosophical Transactions of the Royal Society of London Series В 361, 163-171.
- Li W.K.W., McLaughlin F.A., Lovejoy C. & Carmack E. 2009. Smallest algae thrive as the Arctic Ocean freshens. Science 326, 539.
- Loeng H. 1991. Features of the physical oceanographic conditions of the Barents Sea. Polar Research 10, 5-18.
- Lovejoy C., Massana R. & Pedros-Alio C. 2006. Diversity and ditribution of marine microbial eukaryotes in the Arctic Ocean and adjacent seas. Applied and Environmental Microbiology 72, 3085-3095.
- Lowom J.R., Richman S.E. & Grebmeier J.M. 2003. Diet and body condition of spectacled eiders wintering in pack ice of the Bering Sea. Polar Biology 26, 259-267.
- Lowry L., Kovacs K. & Burkanov V. 2008. Odobenus rosmarus. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet at www.iucnredlist.org on 3 July 2014
- Lu P., Li Z.J., Zhang Z.H. & Dong X.L. 2008. Aerial observations of floe size distribution in the Marginal Ice Zone of summer Prydz Bay. Journal of Geophysical Research—Oceans 113, C02011, doi: 10.1029/2006JC003965.
- Mann K.H. & Lazier J.R.N. 1996. Dynamics of marine ecosystems. Biological-physical interactions in the oceans. 2nd edn. Oxford: Blackwell Science.
- Maslanik J., Stroeve J., Fowler C.W & Emery W. 2011. Distribution and trends in Arctic sea ice age through spring 2011. Geophysical Research Letters 38, LI3502, doi: 10.1029/ 2011GL047735.
- Matrai P.A., Olson E., Suttles S., Hill V., Codispoti L.A., Light B. & Steel M. 2013. Synthesis of primary production in the Arctic Ocean: I. Surface waters, 1954-2007. Progress in Oceanography 110, 93-106.
- McCann K.S. 2000. The diversity-stability debate. Nature 405, 228-233.
- McClelland J.W., Dery S.J., Peterson B.J., Holmes R.M. & Wood E.F. 2006. A pan-Arctic evaluation of changes in river discharge during the latter half of the 20th century. Geophysical Research Letters 33, L06715, doi: 10.1029/ 2006GL025753.
- McGill B.J., Enquist B.J., Weiher E. & Westoby M. 2006. Rebuilding community ecology from functional traits. Trends in Ecology and Evolotion 21, 178-185.
- McPhee M.G., Morison J.H. & Nilsen F. 2008. Revisiting heat and salt exchange at the ice-ocean interface: ocean flux and modeling considerations. Journal of Geophysical Research— Oceans 113, C06014, doi 10.1029/2007JC004383.
- Megrey B.A. & Aydin K.Y. 2009. A macrodescriptor perspective of ecological attributes for the Bering and Barents seas. Deep-Sea Research Part II56, 2132-2140.
- Miyazaki N. 2002. Ringed, Caspian, and Baikal seals Pusa hispida, P. caspica, and P. sibirica. In WF. Perrin et al. (eds.): Encyclopedia of marine mammals. Pp. 1033-1037. San Diego, CA: Academic Press.
- Moore S.E., Grebmeier J.M. & Davies J.R. 2003. Gray whale distribution relative to forage habitat in the northern Bering Sea: current conditions and retrospective summary. Canadian Journal of Zoology 81, 734-742.
- Moore S.E. & Huntington H.P. 2008. Arctic marine mammals and climate change: impacts and resilience. Ecological Applications 18, 157-165.
- Moore S.E., Logerwell E., Eisner L., Farley E.V. Jr., Harwood L.A., Kuletz K., Lowom J., Murphy J.R. & Quakenbush L.T. 2014. Marine fishes, birds and mammals as sentinels of ecosystem variability and reorganization in the Pacific Arctic region. In J.M. Grebmeier & W. Maslowski (eds.): Ecosystem status and trends in a rapidly changing environment. Pp. 337- 392. Dordrecht, Netherlands: Springer.
- Moran S.B., Lomas M.W., Kelly R.P., Gradinger R., Iken K. & Mathis J.T. 2012. Seasonal succession of net primary productivity, particulate organic carbon export, and autotrophic community composition in the eastern Bering Sea. Deep-Sea Research Part II65-70, 84-97.
- Mueter F.J. & Litzow M.A. 2008. Sea ice retreat alters the biogeography of the Bering Sea continental shelf. Ecological Applications 18, 309-320.
- Midler-Navarra D.C. 2008. Food web paradigms: the biochemical view on trophic interactions. International Review of Hydrobiology 93, 489-505.
- Mundy C.J., Gosselin M., Gratton Y., Brown K., Galindo V, Campbell K., Levasseur M., Barber D., Papakyriakou T. & Belanger S. 2014. Role of environmental factors on phytoplankton bloom initiation under landfast sea ice in Resolute Passage, Canada. Marine Ecology Progress Series 497, 39-49.
- Nelson R.J., Ashjian C.J., Bluhm B.A., Conlan K.E., Gradinger R.R., Grebmeier J.M., Hill V.J., Hopcroft R.R., Hunt B.P.V., Joo H.M., Kirchman D.L., Kosobokova K.N., Lee S.H., Li W.K.W., Lovejoy C., Poulin M., Sherr E. & Young K.V. 2014. In J.M. Grebmeier & W. Maslowski (eds.): Ecosystem status and trends in a rapidly changing environment. Pp. 269-336. Dordrecht, Netherlands: Springer.
- Nelson R.J., Carmack E., McLaughlin F.A. & Cooper G.A. 2009. Penetration of Pacific zooplankton into the western Arctic Ocean tracked with molecular population genetics. Marine Ecology Progress Series 381, 129-138.
- Neutel A.M., Heesterbeek J.A.P. & de Ruiter PC. 2002. Stability in real food webs: weak links in long loops. Science 296, 1120-1123.
- Nicolaus M., Katlein C., Maslanik J. & Hendricks S. 2012. Changes in Arctic sea ice result in increasing light transmittance
and absorption. Geophysical Research Letters 39, L24501, doi: 10.1029/2012GL053738. - O'Connor M.I., Bruno J.F., Gaines S.D., Halpern B.S., Lester S.E., Kinlan B.P. & Weiss J.M. 2007. Temperature control of larval dispersal and the implications for marine ecology, evolution, and conservation. Proceedings of the National Academy of Sciences of the United States of America 104, 1-6.
- Overland J.E. & Stabeno P.J. 2004. Is the climate of the Bering Sea wanning and affecting the ecosystem? Eos, Transactions of the American Geophysical Union 85, 309-310.
- Palmer M.A., Saenz B.T. & Anigo K.R. 2014. Impacts of sea ice retreat, thinning, and melt-pond proliferation on the summer phytoplankton bloom in the Chukchi Sea, Arctic Ocean. Deep-Sea Research Part II 105, 85-104.
- Parkinson C.L. & Comiso J.C. 2013. On the 2012 record low Arctic sea ice cover: combined impact of preconditioning and an August storm. Geophysical Research Letters 40, 1356- 1361.
- Pauly D., Christensen V, Guenette S., Pitcher T.J., Sumaila 2002 U. , Walters C.J., Watson R. & Zeller D. 2002. Towards sustainability in world fisheries. Nature 418, 689-695.
- Pauly D., Christensen V. & Walters C. 2000. Ecopath, Ecosim, and Ecospace as tools for evaluating ecosystem impact of fisheries. ICES Journal of Marine Science 57, 697-706.
- Payet J.P. & Suttle C. 2013. To kill or not to kill: the balance between lytic and lysogenic viral infection is driven by trophic status. Limnology and Oceanography 58, 465-474.
- Pearson TH. & Rosenberg R. 1978. Macrobenthic succession in relation to organic enrichment and pollution of the marine environment. Oceanography and Marine Biology—an Annual Review 16, 229-311.
- Pedersen G. & Zeller D. 2001. A mass balance model for the West Greenland marine ecosystem. In S. Guenette et al. (eds.): Fisheries impacts on North Atlantic ecosystems: models and analyses. Pp. 111-127. Vancouver: Fisheries Centre, University of British Columbia.
- Perovich D.K., Ticker W.B., III. & Krishfield R.A. 1989. Oceanic heat flux in the Fram Strait measured by a drifting buoy. Geophysical Research Letters 16, 995-998.
- Peterson B.J., Holmes R.M., McClelland J.W, Vorosmarty C.J., Lammers R.B., Shiklomanov A.I. & Rahmstorf I.A.S.S. 2002. Increasing river discharge to the Arctic Ocean. Science 298, 2171-2173.
- Peterson B.J., McClelland J., Curry R., Holmes R.M., Je W. & Aagaar K. 2006. Trajectory shifts in the Arctic and Subarctic freshwater cycle. Science 313, 1061-1066.
- Piechura J. & Walczowski W. 2009. Wanning of the West Spitsbergen Cunent and sea ice north of Svalbard. Oceanologia 51, 149-164.
- Piepenburg D., Archambault P., Ambrose WG. Jr., Blanchard A., Bluhm B., Carroll C.L., Conlan K.E., Cusson M., Feder, Grebmeier J.M., Jewett S.C., Levesque M., Petryashev, Sejr M.K., Sirenko B.I. & Wlodarska-Kowalczuk M. 2011. Towards a pan-Arctic inventory of the species diversity of the macro- and megabenthic fauna of the Arctic shelf seas. Marine Biodiversity 41, 51-70.
- Piepenburg D., Blackbum TH., von Dorrien C.F., Gutt J., Hall P.O., Hulth S., Kendall M.A., Opalinski K.W., Rachor E. & Schmid M.K. 1995. Partitioning of benthic community respiration in the Arctic (northwestern Barents Sea). Marine Ecology Progress Series 118, 199-214.
- Piwosz K., Spich K., Calkiewicz J., Weydmann A., Kubiszyn A.M. & Wiktor J.M. 2015. Distribution of small phytoflagellates along an Arctic fjord transect. Environmental Microbiology, doi: 10.1111/1462-2920.12705.
- Piwosz K., Walkusz W, Hapter R., Wieczorek P., Hop H. & Wiktor J. 2009. Comparison of productivity and phytoplankton in a warm (Kongsfjorden) and cold (Homsund) Spitsbergen fjord in mid-summer 2002. Polar Biology 32, 549-559.
- Piwosz K., Wiktor J.M., Niemi A., Tatarek A. & Michel C. 2013. Mesoscale distribution and functional diversity of picoeukaryotes in the first-year sea ice of the Canadian Arctic. The ISME Journal 7, 1461-1471.
- Polyakov I.V., Timokhov L.A., Alexeev V.A., Bacon S., Dimitrenko LA., Fortier L., Frolov I.E., Gascard J.C., Hansen E., Ivanov V.V., Laxon S., Mauritzen C., Perovich D., Shimada K., Simmons H.L., Sokolov V.T., Steele M. & Toole J. 2010. Arctic Ocean warming contributes to reduced polar cap. Journal of Physical Oceanography 40, 2743-2756.
- Polyakov I.V., Walsh J.E. & Kwok R. 2012. Recent changes of Arctic multiyear sea ice coverage and the likely causes. Bulletin of the American Meteorological Society 93, 145-151.
- Popova E.E., Yool A., Coward A.C., Dupont F., Deal C., Elliott S., Hunke E., Jin M., Steele M. & Zhang J. 2012. What controls primary production in the Arctic Ocean? Results from an intercomparison of five general circulation models with biogeochemistry. Journal of Geophysical Research— Oceans 117, C00D12, doi 10.1029/2011JC007112.
- Raymond P.A., McClelland J.W, Holmes R.M., Zhulidov A.V., Mull K., Peterson B.J., Striegl R.G., Aiken G.R. & Gurtovaya TY. 2007. Flux and age of dissolved organic carbon exported to the Arctic Ocean: a carbon isotopic study of the five largest Arctic rivers. Global Biogeochemical Cycles 21, GB4011, doi: 10.1029/2007GB002934.
- Reilly S.B., Bannister J.L., Best P.B., Brown M., Brownell R.L. Jr., Butterworth D.S., Clapham P.J., Cooke J., Donovan G.P., Urban J. & Zerbini A.N. 2008a. Balaenoptera acutorostrata. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet at www.iucnredlist.org on 3 July 2014
- Reilly S.B., Bannister J.L., Best P.B., Brown M. Brownell R.L. Jr., Butterworth D.S., Clapham P.J., Cooke J., Donovan G.P., Urban J. & Zerbini A.N. 2008b. Eschrichtius robustus. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet on www.iucnredlist.org on 3 July 2014
- Reilly S.B., Bannister J.L., Best P.B., Brown M., Brownell R.L. Jr., Butterworth D.S., Clapham P.J., Cooke J., Donovan G.P., Urban J. & Zerbini A.N. 2012. Balaena mysticetus. In: IUCN red list of threatened species. Version 2014.1. Accessed on the internet on www.iucnredlist.org 3 July 2014
- Renaud P.E., Berge J., Varpe 0., Loime O.J., Nahrgang J., Ottesen C. & Hallanger I. 2012. Is the poleward expansion
by Atlantic cod and haddock threatening native polar cod, Boreogadus saida? Polar Biology 35, 401-412. - Renaud P.E., Carroll M.L. & Ambrose W.G. Jr. 2008. Effects of global warming on Arctic sea-floor communities and its consequences for higher trophic levels. In C.M. Duarte (ed.): Impacts of global warming on polar ecosystems. Pp. 139- 175. Bilbao: Fundacidn BBVA.
- Renaud P.E., Morata N., Ambrose W.G. Jr., Bowie J.J. & Chiuchiolo A. 2007. Carbon cycling by seafloor communities on the eastern Beaufort Sea shelf. Journal of Experimental Marine Biology and Ecology 349, 248-260.
- Renaud P.E., Morata N., Carroll C.L., Denisenko S.G. & Reigstad M. 2008. Pelagic-benthic coupling in the western Barents sea: processes and time scales. Deep-Sea Research Part II 55, 2372-2380.
- Renaud P.E., Tessmann M., Evenset A. & Christensen G.N. 2011. Benthic food-web structure of an Arctic fjord (Kongsfjorden, Svalbard). Marine Biology Research 7, 13-26.
- Rich J., Gosselin M., Sherr E., Sherr B. & Kirchman D.L. 1997. High bacterial production, uptake and concentrations of dissolved organic matter in the Central Arctic Ocean. Deep- Sea Research Part II44, 1645-1663.
- Rosen D.A.S. & Trites A.W 2000. Pollock and the decline of Steller sea lions: testing the junk-food hypothesis. Canadian Journal of Fisheries and Aquatic Sciences 78, 1243-1250.
- Rubao J., Meibing J. & Varpe O. 2013. Sea ice phenology and timing of primary production pulses in the Arctic Ocean. Global Change Biology 19, 734-741.
- Rysgaard S., Kuhl M., Glud R.N. & Hansen J.W 2001. Biomass, production and horizontal patchiness of sea ice algae in a High-Arctic fjord (Young Sound, NE Greenland). Marine Ecology Progress Series 223, 15-26.
- Sakshaug E. 2004. Primary and secondary production in the Arctic Seas. In R. Stein & R.W. Macdonald (eds.): The organic carbon cycle in the Arctic Ocean. Pp. 57-81. New York: Springer.
- Sakshaug E., Johnsen G., Kristiansen S., von Quillfeldt C., Rey 2019., Slagstad D. & Thingstad F. 2009. Phytoplankton and primary production. In E. Sakshaug et al. (eds.): Ecosystem Barents Sea. 167-208. Trondheim: Tapir Academic Press.
- Sarmento H., Montoya J.M., Vazquez-Dominquez E., Vague D. & Gasol J.M. 2010. Warming effects on marine microbial food web processes: how far can we go when it comes to predictions? Philosophical Transactions of the Royal Society of London В 365, 2137-2149.
- Scott C.L., Falk-Petersen S., Gulliksen B., Tonne O.J. & Sargent J.R. 2001. Lipid indicators of the diet of Gammarus wilkitzkii in the Marginal Ice Zone and in open waters of Svalbard. Polar Biology 24, 575-576.
- Scott C.L., Falk-Petersen S., Sargent J.R., Hop H., Tonne O.J. & Poltermann M. 1999. Lipids and trophic interactions of ice fauna and pelagic zooplankton in the Marginal Ice Zone of the Barents Sea. Polar Biology 21, 65-70.
- Screen J.A. & Simmonds I. 2010. The central role of diminishing sea ice in recent Arctic temperature amplification. Nature 464, 1334-1337.
- Serreze M.C., Barret A.P., Stroeve J., Kindig D.N. & Holland M.M. 2009. The emergence of surface-based Arctic amplification. The Cryosphere 3, 11-19.
- Sherr E.B., Sherr B.F. & Hartz A.J. 2009. Microzooplankton grazing impact in the western Arctic Ocean. Deep-Sea Research Part II56, 1264-1273.
- Shiklomanov A.I. & Lammers R.B. 2009. Record Russian river discharge in 2007 and the limits of analysis. Environmental Research Letters 4, 045015, doi: 10.1088/1748-9326/4/4/ 045015.
- Shimada K., Kamoshida T, Itoh M., Nishino S., Carmack E., McLaughlin F., Zimmermann S. & Proshutinsky A. 2006. Pacific Ocean inflow: influence on catastrophic reduction of sea ice cover in the Arctic Ocean. Geophysical Research Letters 33, L08605, doi: 10.1029/2005GL025624.
- Sirenko B.I. & Gagaev S.Y. 2007. Unusual abundance of macrobenthos and biological invasions in the Chukchi Sea. Russian Journal of Marine Biology 33, 355-364.
- Sirenko B.I. & Kolutin V.M. 1992. Characteristics of benthic biocenoses of the Chukchi and Bering seas. In P.A. Nagel (ed.): Results of the third joint US-USSR Bering & Chukchi Seas Expedition (BERPAC), Summer 1988. Pp. 251-258. Washington, DC: US Fish and Wildlife Service.
- Slagstad D., Ellingsen I.H. & Wassmann P. 2011. Evaluating primary and secondary production in an Arctic Ocean void of summer sea ice: an experimental simulation approach. Progress in Oceanography 90, 117-131.
- Soltwedel T, Bauerfeind E., Bergmann M., Budaeva N., Hoste E., Jaeckisch N., von Juterzenka K., Matthiessen J., Mokievsky V, Nothig E.M., Queric N.V., Sablotny B., Sauter E., Schewe I., Urban-Malinga B., Wegner J., Wlodarska-Kowalczuk M. & Klages M. 2005. HAUSGARTEN— multidisciplinary investigations at a deep-sea long-term observatory in the Arctic Ocean. Oceanography 18, 46-61.
- Soreide J.E., Hop H., Carroll M.L., Falk-Petersen S. & Hegseth E.N. 2006. Seasonal food web structures and sympagic- pelagic coupling in the European Arctic revealed by stable isotopes and a two- source food web model. Progress in Oceanography 71, 59-87.
- Soreide J.E., Leu E., Berge J., Graeve M. & Falk-Petersen S. 2010. Timing of blooms, algal food quality and Calanus glacialis reproduction and growth in a changing Arctic. Global Change Biology 16, 3154-3163.
- Springer A.M., McRoy C.P. & Flint M.V. 1996. The Bering Sea green belt: shelf-edge processes and ecosystem production. Fisheries Oceanography 5, 205-223.
- Squire V.A. 2007. Of ocean waves and sea-ice revisited. Cold Regions Science and Technology 49, 110-133.
- Squire V.A., Dugan J.P., Wadhams P., Rottier P.J. & Liu A.K. 1995. Of ocean waves and sea ice. Annual Review of Fluid Mechanics 27, 115-168.
- Steele M., Ermold W. & Zhang J.L. 2008. Arctic Ocean surface warming trends over the past 100 years. Geophysical Research Letters 35, L02614, doi: 10.1029/2007GL031651.
- Stocker T.F., Qin D., Planner G.-K., Hgnor M., Allen S.K., Boschung J., Nauels A., Xia Y., Вех V. & Midgley P.M. (eds.): 2013. Climate change 2013. The physical science basis. Contribution
of Working Group I to the fifth assessment report of the Intergovernmental Panel on Climate Change. Cambridge: Cambridge University Press. - Strong C. & Rigor LG. 2013. Arctic Marginal Ice Zone trending wider in summer and narrower in winter. Geophysical Research Letters 40, 4864-4868.
- Sturluson M., Gissel Nielsen T. & Wassmann P. 2008. Bacterial abundance, biomass and production during spring blooms in the northern Barents Sea. Deep-Sea Research Part II 55, 2186-2198.
- Symon C., Arris L. & Heal B. 2005. Arctic climate impact assessment. Cambridge: Cambridge University Press.
- lamelander T., Reigstad M., Hop H., Carroll M.L. & Wassmann P. 2008. Pelagic and sympagic contribution of organic matter to zooplankton and vertical export in the Barents Sea Marginal Ice zone. Deep-Sea Research Part II55, 2330-2339.
- lamelander T., Renaud P.E., Hop H., Carroll M.L., Ambrose WG. Jr. & Hobson K.A. 2006. Trophic relationships and pelagic-benthic coupling during summer in the Barents Sea Marginal Ice Zone, revealed by stable carbon and nitrogen isotope measurements. Marine Ecology Progress Series 310, 33-46.
- Timmermans M.L. & Winsor P. 2013. Scales of horizontal density structure in the Chukchi Sea surface layer. Continental Shelf Research 52, 39-46.
- Tremblay J.E., Simpson K., Martin J., Miller L., Gratton Y., Barber D. & Price N.M. 2008. Vertical stability and the annual dynamics of nutrients and chlorophyll fluorescence in the coastal, southeast Beaufort Sea. Journal of Geophysical Research—Oceans 113, C07S90, doi: 10.1029/2007JC004547.
- Trites A.W, Livingston P.A., Vasconcellos M.C., Mackinson S., Springer A.M. & Pauly D. 1999. Ecosystem considerations and the limitations of ecosystem models in fisheries management: insights from the Bering Sea. Ecosystem Approaches for Fisheries Management, Alaska Sea Grant College Program, Report 99-01. Fairbanks: University of Alaska Sea Grant.
- Vander Zanden M.J., Casselman J.M. & Rasmussen J.B. 1999. Stable isotope evidence for the web consequences of species invasions in lakes. Nature 401, 464-467.
- Van Oevelen D., Bergmann M., Soetaert K., Bauerfeind E., Hasemann C., Klages M., Schewe I., Soltwedel T. & Budaeva N. 2011. Carbon flows in the benthic food web at the deep- sea observatory HAUS GARTEN (Fram Strait). Deep-Sea Research Part 158, 1069-1083.
- Van Oevelen D., Van den Meers die K., Meysman F.J.R., Soetaert K., Middleburg J.J. & Vezina A.F. 2010. Quantifying food web flows using linear inverse models. Ecosystems 13, 32-45.
- Vanreusel A., Clough L.M., Jacobsen K., Ambrose W.G. Jr., Jivaluk J., Ryheul V, Herman R. & Vincx M. 2000. Meiobenthos of the central Arctic Ocean with special emphasis on the nematode community structure. Deep-Sea Research Part 147, 1855-1879.
- Vaquer-Sunyer R., Duarte C.M., Santiago R., Wassmann P. & Reigstad M. 2010. Experimental evaluation of planktonic respiration response to warming in the European Arctic Sector. Polar Biology 33, 1661-1671.
- Varpe O. 2012. Fitness and phenology: annual routines and zooplankton adaptations to seasonal cycles. Journal of Plankton Research 34, 267-276.
- Wadhams P. 2000. Ice in the ocean. Amsterdam: Gordon & Breach Science Publishers.
- Wadhams P, Squire V.A., Goodman D.J., Cowan A.M. & Moore S.C. 1988. The attenuation rates of ocean waves in the Marginal Ice Zone. Journal of Geophysical Research 93, 6799-6818.
- Walczowski W. & Piechura J. 2006. New evidence of warming propagating toward the Arctic Ocean. Geophysical Research Letters 33, L12601, doi: 10.1029/2006GL025872.
- Walczowski W, Piechura J., Goszczko I. & Wieczorek P. 2012. Changes in Atlantic water properties: an important factor in the European Arctic marine climate. ICES Journal of Marine Science 69, 864-869.
- Walsh J.J., McRoy C.P., Coachman L.K., Goering J.J., Nihoul J. J., Whitledge T.E., Blackbum Т.Н., Parker P.L., Wirick C.D., Shuert P.G., Grebmeier J.M., Springer A.M., Tripp R.D., Hansell D.A., Djenidi S., Deleersnijder E., Henriksen K., Lund B.A., Andersen P., Muller-Karger F.E. & Dean K. 1989. Carbon and nitrogen cycling within the В ering/Chukchi seas: source regions of organic matter affecting AOU demands of the Arctic ocean. Progress in Oceanography 22, 279-361.
- Walther G.R., Post E., Convey P., Menzel A., Parmesank C., Beebee T.J.C., Fromentin J.M., Hoegh-Guldberg O. & Bairlein F. 2002. Ecological responses to recent climate change. Nature 416, 389-395.
- Wanless S., Harris M., Redman P. & Speakman J. 2005. Low energy values of fish as a probable cause of a major seabird breeding failure in the North Sea. Marine Ecology Progress Series 294, 1-8.
- Wassmann P. 2006. Structure and function of contemporary food webs on Arctic shelves: an introduction. Progress in Oceanography 71, 123-128.
- Wassmann P, Carroll J. & Bellerby R.G.J. 2008. Carbon flux and ecosystem feedback in the northern Barents Sea in an era of climate change: an introduction. Deep-Sea Research Part II 55, 2143-2153.
- Wassmann P., Duarte C.M., Agusti S. & Sejr M.K. 2011. Footprints of climate change in the Arctic marine ecosystem. Global Change Biology 17, 1235-1249.
- Wassmann P. & Reigstad M. 2011. Future Arctic Ocean seasonal ice zones and implications for pelagic-benthic coupling. Oceanography 24, 220-231.
- Wassmann P., Reigstad M., Haug T., Rudels B., Carroll M.L., Hop H., Gabrielsen G.W, Falk-Petersen S., Denisenko S.G., Arashkevich E., Slagstad D. & Pavlova O. 2006. Food webs and carbon flux in the Barents Sea. Progress in Oceanography 71, 232-287.
- Wassmann P., Slagstad D. & Ellingsen I. 2010. Primary production and climatic variability in the European sector of the Arctic Ocean prior to 2007: preliminary results. Polar Biology 33, 1641-1650.
- Wassmann P., Slagstad D., Riser C.W & Reigstad M. 2006. Modelling the ecosystem dynamics of the Barents Sea
including the Marginal Ice Zone: II. Carbon flux and interannual variability. Journal of Marine Systems 59, 1-24. - Weeks WF. 2010. On sea ice. Fairbanks, AK: University of Alaska Press.
- Wells L.E. & Deming J.W 2006. Modelled and measured dynamics of viruses in Arctic winter sea-ice brines. Environmental Microbiology 8, 1115-1121.
- Werner I. & Auel H. 2005. Seasonal variability in abundance, respiration and lipid composition of Arctic under-ice amphipods. Marine Ecology Progress Series 292, 251-262.
- Werner I., Auel H. & Friedrich C. 2002. Carnivorous feeding and respiration of the Arctic under-ice amphipod Gammarus wilkitzkii. Polar Biology 25, 523-530.
- Wfslawski J.M., Kwasniewski S. & Stempniewicz L. 2009. Warming in the Arctic may result in the negative effects of increased biodiversity. Polarforschung 78, 105-108.
- Wheeler P.A., Watkins J.M. & Hansing R.L. 1997. Nutrients, organic carbon and nitrogen in the upper water column of the Arctic Ocean: implications for the sources of dissolved organic carbon. Deep-Sea Research Part II44, 1571-1592.
- Whitehouse G.A., Aydin K., Essington T.E. & Hunt G.L. Jr. 2014. A trophic mass balance model of the eastern Chukchi Sea with comparisons to other high-latitude systems. Polar Biology 37, 911-939.
- Wienerroither R.M., Nedreaas K., Uiblein F., Christiansen J.S., Byrkjedal I. & Karamushko O. 2011. The marine fishes of Jan Mayen Island, NE Atlantic—past and present. Marine Biodiversity 41, 395-411.
- Wilson W.H., Tarran G.A., Schroeder D., Cox M.J., Oke J. & Malin G. 2002. Isolation of viruses responsible for the demise of an Emiliania huxleyi bloom in the English Channel. Journal of the Marine Biological Association of the United Kingdom 82, 369-377.
- Woodgate R.A., Aagaard K. & Weingartner T. 2006. Interannual changes in the Bering Strait fluxes of volume, heat and freshwater between 1991 and 2004. Geophysical Research Letters 33, L15609, doi: 10.1029/2006GL026931.
- Woodgate R.A., Weingartner T. & Lindsay R. 2010. The 2007 Bering Strait oceanic heat flux and anomalous Arctic sea-ice retreat. Geophysical Research Letters 37, L01602, doi: 10.1029/ 2009GL041621.
- Woodgate R.A., Weingartner T. & Lindsay R. 2012. Observed increases in Bering Strait oceanic fluxes from the Pacific to the Arctic from 2001 to 2011 and their impacts on the Arctic Ocean water column. Geophysical Research Letters 39, L24603, doi: 10.1029/2012GL054092.
- Zhang J., Spitz Y.H., Steele M., Ashijian C., Campbell R., В eline L. & Matrai P. 2010. Modeling the impact of declining sea ice on the Arctic marine planktonic ecosystem. Journal of Geophysical Research—Oceans 115, C10015, doi: 10.1029/ 2009JC005387.





